
Introduction
Specifying borosilicate glass without understanding its thermal thresholds is a recurring mistake in pharmaceutical, food processing, and chemical applications — one that leads to under-designed thermal protection, wrong component selection, and failures that are entirely preventable.
As an amorphous material, borosilicate glass transitions through a series of distinct thermal thresholds — softening, working, and full melting — rather than hitting a single sharp melt temperature the way crystalline materials do. Each threshold carries different engineering implications, and confusing them leads to incorrect component selection.
The full melting range (approximately 1,252–1,650°C) is an absolute ceiling that matters only during glass manufacturing. For process engineers, the actionable numbers are the softening point (~820°C) and the maximum continuous working temperature (~300–500°C) — the two values that determine whether borosilicate glass is the right material for a given application.
TL;DR
- Borosilicate melts at 1,252–1,650°C depending on grade, but its safe continuous working limit is only 300–500°C
- Its low thermal expansion coefficient (3.3 × 10⁻⁶ /K) drives thermal shock resistance—not the melting point
- Softening point (~820°C), working temperature (~500°C), and melting point are three distinct thresholds—confusing them leads to equipment failure
- Exceeding the rated working temperature causes deformation, seal failure, and dimensional loss before the softening point is ever reached
- USP/EP Type I chemical resistance, electrical insulation, and optical clarity extend borosilicate's value well past thermal applications
What the Melting Point of Borosilicate Glass Represents
The melting point of borosilicate glass is the temperature at which its amorphous silica-boron oxide network transitions from a rigid solid to a viscous, fully fluid state. Because borosilicate is amorphous rather than crystalline, this occurs progressively over a temperature range rather than at a fixed point. The working point is defined at 1,252–1,260°C (viscosity of 10⁴ poise), while the upper melting temperature reaches approximately 1,650°C for full fluidity during manufacturing operations.
This melting point functions as an upper absolute limit, not an operating parameter. It defines the ceiling of the material's thermal envelope. The glass's practical utility is governed by the softening point (~820°C) and working temperature (300–500°C), which are significantly lower.
Comparative Glass Performance:
| Glass Type | Softening Point (°C) | Working Point (°C) | CTE (× 10⁻⁶/K) |
|---|---|---|---|
| Borosilicate 3.3 | 821–825 | 1,252–1,260 | 3.25–3.3 |
| Soda-lime | 720 | 1,040 | 9.1 |
| Fused silica | 1,590 | Not listed | 0.52–0.57 |

Borosilicate occupies a useful middle ground: it outperforms soda-lime glass thermally while remaining far easier to form than fused silica, which costs substantially more to process. That tradeoff makes it the default choice for sight glasses, process tubing, and laboratory vessels where both thermal shock resistance and fabricability matter.
For engineers specifying process components, the melting point is a manufacturing constraint — it matters for lampworking, tube drawing, and molding, not for service conditions. In service, the softening point and maximum continuous working temperature are what determine whether a component holds up.
Composition and Its Role in Defining the Melting Point
Borosilicate glass is composed primarily of silica (SiO₂, ~65–85%) and boron trioxide (B₂O₃, ~8–15%), with smaller amounts of sodium oxide, aluminum oxide, and potassium oxide. B₂O₃ modifies the glass network by substituting some Si-O bonds with B-O bonds. This substitution lowers viscosity at elevated temperatures and shifts both softening and melting behavior relative to pure silica.
Research confirms that B₂O₃ "can lower the viscosity of the glass melt, facilitating easier processing" and can significantly reduce the melting temperature relative to pure silica. B₂O₃ itself has a melting point of only 450°C; when incorporated into silicate glass, it substantially reduces processing temperatures.
Different borosilicate families exhibit meaningfully different thermal profiles:
- Non-alkaline-earth (3.3 expansion): SiO₂ >80%, B₂O₃ 12–13%, softening point ~825°C—the most chemically resistant, used in laboratory and pharmaceutical contexts
- Alkaline-earth (4.9–5.4 expansion): SiO₂ ~75%, B₂O₃ 8–12%—used for pharmaceutical tubing and ampoules where forming properties are prioritized
- High-borate (5.0 expansion): SiO₂ 65–70%, B₂O₃ 15–25%, lower softening points—used for metal-sealing applications but with reduced chemical resistance
Thermal Operating Range: From Softening Point to Full Melt
Specifying borosilicate glass for any engineered system requires understanding at least three distinct temperature thresholds: the maximum continuous working temperature, the softening point, and the melting point. Mixing up these values is one of the most common and consequential specification errors in process component selection.
Nominal Operating Range
Standard borosilicate glass has a maximum continuous working temperature of approximately 300–500°C, depending on grade and product form. The softening point sits at approximately 820–821°C for type 7740/Pyrex-grade glass. These values assume:
- Uniform heating
- No rapid thermal cycling
- No mechanical stress concentrations
- Correct grade selection
The working temperature listed on most datasheets assumes stable, continuous exposure—not peak or transient conditions. Real-world duty cycles, thermal gradients, and assembly constraints reduce the practical safe operating margin.
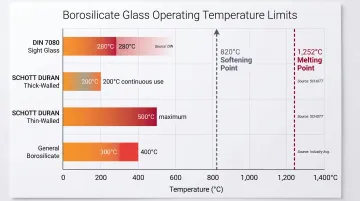
Operating Limits by Application Context:
| Specification Context | Temperature Limit | Source |
|---|---|---|
| DIN 7080 (borosilicate sight glass) | 280°C | Industry standard |
| SCHOTT DURAN (thick-walled components) | 200°C continuous | Manufacturer specification |
| SCHOTT DURAN (thin-walled, shape stability) | 500°C maximum | Manufacturer specification |
| General borosilicate | 300–400°C | Material datasheets |
Artesian Systems' tri-clamp borosilicate sight glasses (1.5"–6" range) are rated for pharmaceutical-grade service at temperatures from -40°C to 500°C, with compliance to FDA 21CFR177.2600/1550, 3A Sanitary Standards, and USDA approval—demonstrating the upper end of continuous working capability for properly engineered components.
Allowable Tolerance and Boundary Limits
The softening point is defined as the temperature at which viscosity drops to approximately 10⁷·⁶ poise. At this point, borosilicate glass begins to deform under its own weight or applied load, making this the practical upper limit for structural integrity in most component applications.
Distinguish between:
- Transient excursions a properly annealed component can survive without fracture (short-term exposure)
- Sustained operating conditions within which dimensional stability and material properties hold (continuous exposure)
The strain point (~510°C for Corning 7740) marks "the maximum value for short-term heat load." Above the strain point, permanent deformation and residual stress accumulation begin.
Safe Operating Margin
Borosilicate glass accumulates thermal stress when temperature gradients develop across its cross-section. The low coefficient of thermal expansion (~3.3 × 10⁻⁶ /K) reduces this stress compared to soda-lime glass, but does not eliminate it—particularly in thick-walled components or assemblies with constrained joints.
Borosilicate glass typically withstands approximately 165–170°C temperature differential before fracture risk increases significantly, compared to soda-lime glass's tolerance of only ~40°C. In sterilization, CIP/SIP cycles, or rapid-transition applications, engineers should build in explicit thermal margin rather than operating near this threshold.
For cyclic thermal service, wall thickness must be evaluated against pressure requirements. Thicker glass develops larger center-to-surface temperature gradients during heating and cooling — generating higher mechanical stress and greater fracture risk than thin-walled equivalents under the same duty cycle.
Key Material Properties of Borosilicate Glass
The melting point sets the upper thermal limit, but day-to-day performance depends on the full property profile: thermal, mechanical, chemical, electrical, and optical. Each of these governs how borosilicate behaves under real operating conditions.
Property 1: Thermal Expansion and Thermal Shock Resistance
The coefficient of thermal expansion (CTE) for standard borosilicate glass is approximately 3.3 × 10⁻⁶ /K—roughly one-third that of soda-lime glass. This low CTE is the primary driver of borosilicate's thermal shock resistance. Lower expansion means lower stress buildup during rapid temperature changes.
Thermal shock resistance comparison:
- Borosilicate: ~166–170°C temperature differential before fracture
- Soda-lime: ~37°C differential
- Fused quartz: Nearly 10× greater thermal stress tolerance than borosilicate
This property governs material selection for sight glasses, process vessels, and any component subjected to rapid heating or cooling cycles.
Property 2: Mechanical Properties
Key mechanical values for engineering applications:
| Property | Value | Notes |
|---|---|---|
| Mohs Hardness | ~6 | Scratch resistance |
| Knoop Hardness | 420–520 kg/mm² | Surface hardness |
| Young's Modulus | 62.75–64 GPa | Stiffness |
| Poisson's Ratio | 0.20 | Lateral strain response |
| Density | 2.23 g/cm³ | Mass calculation |
Borosilicate glass is significantly stronger in compression than in tension. Surface flaws or scratches disproportionately reduce tensile strength, which makes surface condition a practical requirement for component design, handling protocols, and quality verification of annealing standards.
Property 3: Chemical Resistance and Inertness
Borosilicate glass exhibits high resistance to acids, alkalis, and organic solvents due to its low alkali content and stable silica-boron network, which minimizes ion leaching and surface degradation. This chemical inertness is why it is classified as USP/EP Type I glass for pharmaceutical primary packaging.
Regulatory classifications:
- USP <660> Type I: "Neutral glass" with "high hydrolytic resistance"—ideal for all injectable preparations with acid, neutral, and alkaline pH
- European Pharmacopoeia (Ph. Eur.) 3.2.1 Type I: Equivalent classification
- ASTM E438 Type I, Class A: Low-expansion borosilicate glass for laboratory apparatus
For process engineers specifying components that must meet these classifications, material compliance is not a secondary consideration. Artesian Systems manufactures FDA 21CFR177.2600/1550 and USDA-compliant borosilicate sight glasses and process components for pharmaceutical, food, and chemical processing environments where regulatory traceability is required at the material level.
Property 4: Electrical and Optical Properties
Electrical insulation capability:
- Volume resistivity: >10¹⁵ Ω·m, making borosilicate an effective electrical insulator
- Applications: Electronics, semiconductor fabrication, sensor applications
Optical properties:
- Refractive index: ~1.47–1.54 across the visible range
- Abbe number: ~65 (low optical dispersion)
- Suitable for optical components, instrument lenses, and telescope mirrors where dimensional stability with temperature is critical
Factors That Affect Borosilicate Glass Thermal Performance in Real-World Applications
Published thermal specifications—melting point, softening point, working temperature—are determined under controlled laboratory conditions. Real-world performance deviates from those specs in predictable ways:
- Borosilicate subtype: Grades 3.3, 4.9, and 5.4 expansion have different softening points and working temperatures. Specifying "borosilicate" without the subtype creates performance ambiguity.
- Wall thickness and geometry: Thicker walls produce larger thermal gradients between center and surface. SCHOTT rates DURAN at 200°C maximum for large thick-walled components versus 500°C for thin-walled items.
- Surface condition: Scratches and existing cracks act as stress concentrators under thermal cycling. Glass tensile strength is highly sensitive to surface defects—even minor ones can propagate under repeated thermal load.
- Heating uniformity: Uniform radiant heat produces different stress patterns than localized flame or contact heating. Thermal shadowing—where gasket-protected areas stay cool while viewing areas heat—creates extreme gradients within a single component and can cause failure even within rated temperature limits.
- Annealing quality: Residual stress locked in during manufacturing determines how the component behaves in service. Inadequately annealed glass fractures at lower thermal differentials than published specs suggest; fine annealing brings residual stress to minimal levels.
- Chemical exposure: Concentrated alkali solutions, hydrofluoric acid, and hot phosphate chemistries attack the glass surface, thin the effective wall, and reduce thermal shock threshold over time. This "stress corrosion" creates surface damage that functions as a stress concentrator—a degradation mode absent from static material data sheets.

Implications of Exceeding Thermal and Chemical Operating Limits
When borosilicate glass operates above its continuous working temperature (above ~500°C for standard grades), it approaches the strain point and anneal point range. At these temperatures:
- Internal stresses relax unpredictably
- Dimensional drift occurs in precision components
- Loss of seal integrity occurs in fitted assemblies
- Progressive structural weakening develops — often invisible until catastrophic fracture occurs under subsequent thermal cycling
Thermal Fracture and Process Failure
When rapid thermal cycling exceeds the material's safe differential, fracturing initiates at surface flaws and propagates inward. In process piping or vessel applications, this releases contents and creates safety and contamination risks across pharmaceutical, food, and chemical processing settings.
Preventing these failures requires material selection based on full process duty-cycle analysis — not just peak temperature ratings. For sanitary process systems, that means accounting for CIP/SIP cycles, pasteurization temperatures, and chemical exposure specific to applications like pharmaceutical USP-32 water systems and food processing lines. Artesian Systems applies this duty-cycle approach when engineering custom sanitary systems to ensure components are rated for real operating conditions, not just nominal ones.
Compliance and System Integrity Consequences
For regulated applications in pharma, food, and bioprocessing, material failure or degradation of borosilicate glass components can trigger:
- Regulatory non-conformance events
- Product contamination
- Process downtime
- Validation failures
Specifying components against actual operating conditions, not theoretical peak values, is what keeps regulated processes in conformance. The ~1,650°C melting point reflects what borosilicate glass can withstand during manufacture; the 280°C DIN 7080 sight glass rating reflects what it can reliably sustain in service. Both numbers matter, but only one governs system design.
Common Misinterpretations of Borosilicate Glass Thermal Specifications
The most consequential misinterpretation in borosilicate glass specification is treating the published melting point (~1,252–1,650°C) as the practical operating limit. The actual engineering boundaries are the maximum continuous working temperature (~300–500°C) and the softening point (~820°C) — and conflating these figures leads directly to under-designed thermal protection and incorrect component selection.
DIN 7080 rates borosilicate sight glass to only 280°C for process applications — approximately one-sixth of the commonly cited melting point. SCHOTT specifies 200°C continuous for large, thick-walled DURAN components. That gap is not a safety margin to exploit. These ratings reflect real-world constraints: thermal gradients, pressure loads, and cyclic stress that no published melting point accounts for.
Lab Glass vs. Industrial Process Components
Borosilicate glass cookware and laboratory beakers are built to different standards than engineered process components. Applying the same temperature differential tolerance across all borosilicate products ignores:
- Geometry differences (thick-walled pressure vessels vs. thin-walled beakers)
- Annealing quality (DIN 7080 imposes specific annealing and stress requirements)
- Surface condition (industrial components may have machined surfaces vs. fire-polished lab glass)
- Use profiles (continuous process exposure vs. intermittent laboratory use)
These differences matter in procurement. Some industrial sight glasses on the market fail USP Type I standards, carrying a Type NP (non-parenteral) classification — unsuitable where chemical durability and thermal shock resistance are critical. Specifying "borosilicate glass" by name alone is not enough; procurement documents should call out explicit certifications such as USP Type I, DIN 7080, or 3-A compliance to ensure the component actually meets process requirements.

Conclusion
The melting point of borosilicate glass defines the absolute thermal ceiling of the material—but practical system reliability depends on understanding and respecting the working temperature and softening point: the operationally relevant limits. Published specifications are starting points, not guarantees.
Grade, geometry, annealing quality, surface condition, chemical environment, and heating regime all shift where the real operating limits lie. Apply engineering judgment to actual process conditions rather than nominal datasheet values — that's what keeps components performing reliably over time.
For pharmaceutical, food, and chemical process applications, three specification practices reduce risk:
- Specify non-alkaline-earth borosilicate (3.3 expansion, ASTM E438 Type I Class A, USP/EP Type I) with explicit certification requirements
- Reference component-specific manufacturer datasheets and applicable DIN/ASTM standards — not generic "borosilicate glass" melting point data
- Account for full process duty cycles, including CIP/SIP thermal cycling, chemical exposure, and pressure loads
The full suite of thermal, mechanical, and chemical properties determines whether borosilicate is the right material for a given application. When a process demands capability beyond borosilicate's 500°C working limit, fused silica (quartz glass) is the appropriate step up — though at a meaningfully higher cost.
Frequently Asked Questions
What is the composition of borosilicate glass?
Borosilicate glass is composed primarily of silica (SiO₂, ~65–85%) and boron trioxide (B₂O₃, ~8–15%), with minor amounts of sodium oxide, aluminum oxide, and potassium oxide. This combination produces its characteristically low thermal expansion and high chemical resistance.
Is borosilicate glass resistant to thermal shock?
Yes, borosilicate glass is significantly more resistant to thermal shock than soda-lime glass due to its low coefficient of thermal expansion (~3.3 × 10⁻⁶ /K). It can withstand temperature differentials of approximately 165–170°C without fracturing, compared to about 40°C for standard soda-lime glass—roughly 4.5× greater thermal shock tolerance.
Is borosilicate glass stronger than regular glass?
Borosilicate glass is harder and more thermally durable than standard soda-lime glass, but not unconditionally "stronger." It has comparable or slightly lower tensile strength. Its advantage lies in thermal shock resistance and chemical durability rather than raw mechanical strength alone.
Is borosilicate glass chemically resistant and non-toxic?
Yes, borosilicate glass resists most acids, alkalis, and solvents, leaches minimal ions into contact media, and is non-toxic and BPA-free. It meets USP/EP Type I glass standards—the least reactive pharmaceutical glass classification—making it appropriate for pharmaceutical, food, and medical applications requiring chemical inertness.
Can I pour boiling water into borosilicate glass?
Yes, properly manufactured borosilicate glass handles boiling water without cracking, thanks to its low thermal expansion. Avoid extreme simultaneous temperature shifts—such as moving a boiling vessel directly onto ice—which can exceed even its thermal shock tolerance.
What glass has the highest melting point?
Fused silica (quartz glass) has the highest melting point among common glass types at approximately 1,700°C, followed by borosilicate glass at approximately 1,252–1,650°C depending on grade. Soda-lime glass melts at a lower range of approximately 1,000–1,600°C.


