
Introduction
A single contamination event in pharmaceutical process piping can trigger batch failures, regulatory citations, or costly product recalls. The FDA has documented that nearly all drugs recalled for nonsterility or lack of sterility assurance between 1980–2000 came from aseptic processing lines — meaning filtration failures, not formulation errors, were the root cause. That record makes inline filtration a compliance issue as much as an engineering one.
Inline filtration — filters integrated directly into process piping rather than standalone housings — has become the preferred approach in modern pharmaceutical and bio-pharma facilities. These systems maintain continuous purity, reduce dead-legs that harbor biofilm, and support CIP/SIP cycles critical to sanitary processing.
Below is a practical breakdown of the top inline filtration solutions for pharmaceutical process piping — what each does well, where it fits, and how to match the right solution to your process requirements.
TL;DR
- Inline filtration removes particulates, bioburden, and contaminants directly within the pharmaceutical flow path, maintaining cGMP compliance throughout
- Solutions span sterilizing-grade membrane filters (0.2 µm), depth filters, pleated cartridges, and custom assemblies meeting 3-A, FDA 21 CFR, and USP Class VI standards
- Key selection factors: micron rating, CIP/SIP compatibility, tri-clamp connections, material compliance, and engineering support
- Leading providers include Pall, Sartorius, Parker, Donaldson, and Artesian Systems
- Custom-engineered systems are often the right call for non-standard flow rates, specialty alloys, or unusual piping configurations
What Is Inline Filtration in Pharmaceutical Process Piping?
Inline filtration refers to filter assemblies mounted directly within the process pipe run, allowing continuous removal of particulates, bioburden, or contaminants without diverting flow or shutting down the line. Unlike side-stream or offline systems, inline filters maintain uninterrupted processing while protecting product purity at every point in the piping network.
That protection spans nearly every stage of pharmaceutical manufacturing:
- Sterile liquid preparation and aseptic fill lines: sterilizing-grade filtration where terminal sterilization is not feasible
- WFI distribution loops: bioburden control at point-of-use
- Buffer and media transfer: bioburden reduction before final sterilizing filtration
- Compressed gas and air lines: autoclave air, lyophilizer vacuum breaks, and WFI tank vents
- API synthesis: particulate protection for downstream processes
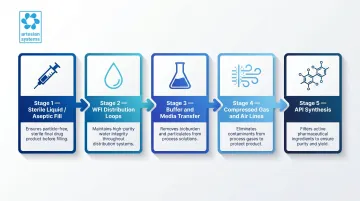
Regulatory guidance reinforces this approach at every stage. EU GMP Annex 1 (2022) recommends "an additional filtration through a sterile sterilising grade filter, as close to the point of fill as possible" as part of contamination control strategies.
Understanding where inline filtration fits — and what each stage demands — is the starting point for choosing the right solution.
Top Inline Filtration Solutions for Pharmaceutical Process Piping
Choosing the wrong inline filter in a pharmaceutical process line isn't just a performance problem — it's a compliance risk. The vendors below represent proven options across sterilizing-grade membrane, depth, and gas filtration, evaluated on regulatory credentials, sanitary construction, and engineering support depth.
Pall Corporation
Pall is a global filtration leader with decades of pharmaceutical and bioprocess experience. Their inline filtration portfolio covers membrane, depth, and sterilizing-grade cartridge filters designed for direct integration into sanitary piping.
Why engineers specify Pall:
- Kleenpak and SUPRAdisc product lines with broad validation data packages
- Extensive FDA and EMA regulatory support documentation
- Compatible with high-purity water systems, sterile bulk drug, and aseptic fill processes
Key specifications:
| Feature | Details | |---------|---------|| | Key Filter Types | Sterilizing-grade membrane (0.2 µm), depth, vent/gas filters; single-use and reusable configurations | | Sanitary Connection Options | Tri-clamp, Sanitary DIN, custom manifold configurations | | Compliance & Certifications | FDA, EMA, cGMP; extensive extractables/leachables data; USP Class VI materials |
Pall's Supor EKV (PES, 0.2 µm) and Bio-Inert (Nylon 6,6, 0.2 µm) membranes are validated per ASTM 838-05 with a bacterial challenge greater than 10^7 CFU/cm² using Brevundimonas diminuta — the industry standard for sterilizing-grade filters.
Sartorius
Sartorius offers inline filtration solutions purpose-built for bioprocess and pharmaceutical piping, including Sartoguard, Sartobran, and Sartopore membrane capsule and cartridge filters for sterile and clarification applications.
Where Sartorius excels:
- High-efficiency PES and PVDF membrane technology
- Strong regulatory filing support (drug master files)
- Integrated automation compatibility (Sartocheck integrity-test-compatible housings)
- Validated scalability from lab to full production — 4.5 cm² Sartoscale units up to 16.2 m² Maxicaps MR
Key specifications:
| Feature | Details | |---------|---------|| | Key Filter Types | PES and PVDF membrane cartridges (0.1–0.45 µm), pleated depth capsules, gas/vent filters | | Sanitary Connection Options | Tri-clamp, Sartocheck integrity-test-compatible housings | | Compliance & Certifications | cGMP, FDA 21 CFR, USP Class VI; drug master file (DMF) support available |
Sartopore 2 filters are validated per ASTM F-838 principles and individually integrity-tested before shipment.
Parker Hannifin – Bioscience Division
Parker's Bioscience Division produces sanitary inline filter housings and cartridges engineered for pharmaceutical and food-grade process piping, emphasizing hygienic design principles — dead-leg-free flow paths and full drainability.
Design strengths:
- Broad range of sanitary housing configurations (single and multi-cartridge)
- CIP/SIP-validated designs
- Compatible with liquid and gas/vent filtration across product contact and utility streams
Key specifications:
| Feature | Details | |---------|---------|| | Key Filter Types | Depth, membrane (PTFE, PES), activated carbon inline cartridges; sanitary gas filters | | Sanitary Connection Options | Tri-clamp (1.5"–4"), Bevel-seat, flanged sanitary connections | | Compliance & Certifications | 3-A Sanitary Standards, FDA-compliant wetted materials, ASME BPE design guidelines |
Parker's PROPOR SG line features PES membranes in 0.1, 0.2, and 0.45 µm ratings with hot water sanitization capability up to 90°C (194°F), supporting CIP protocols without full steam sterilization.
Donaldson Process Filtration
Donaldson offers pharmaceutical-grade inline process filters including Ultra-Web and LifeTec membrane cartridge systems, designed for both liquid and compressed gas filtration in product contact and utility piping.
Operational strengths:
- High-capacity filter media with extended service life
- Compatibility with aggressive cleaning protocols
- Certified 3-A Symbol holder with housings polished to Ra 32 µin
- Suited for high-throughput lines where minimizing filter change-outs reduces downtime
Key specifications:
| Feature | Details | |---------|---------|| | Key Filter Types | Pleated membrane (0.1–1.0 µm), high-flow depth cartridges, compressed air/gas inline filters | | Sanitary Connection Options | Tri-clamp, threaded sanitary, inline gas manifold configurations | | Compliance & Certifications | FDA 21 CFR compliant wetted materials; cGMP documentation packages available |
Donaldson's LifeTec PES-WN achieves LRV 7 (log reduction value) for bacterial retention and is manufactured in a Class 7 cleanroom with individual integrity testing and serial number traceability for all 10" sterile modules.
Artesian Systems
Artesian Systems is a specialized engineering firm with nearly two decades of experience in process pipe engineering for pharmaceutical, bio-pharma, chemical, and food industries. They offer custom-designed inline filtration systems built around solid-state sanitary components and precision-machined specialty alloys.
What sets Artesian apart:
Artesian approaches inline filtration as a full process engineering problem, not a component catalog selection. Their scope includes:
- Complete pipe system design (P&IDs, hydraulic calculations, dead-leg-free configurations)
- Custom component fabrication via CNC machining in specialty alloys (Inconel, Hastelloy, 316L stainless)
- Passivation of wetted components (pre-pickling to enhance chromium oxide layer)
- Prototype system development for processes where off-the-shelf solutions fall short
Their systems are built to FDA 21 CFR 177.2600/1550, 3-A, and USDA compliance standards with ISO9001-approved quality controls.
Key specifications:
| Feature | Details | |---------|---------|| | Key Filter Types | Custom inline filtration screens (5–100 micron) and assemblies; specialty alloy and composite sanitary filter systems; solid-state sanitary components | | Sanitary Connection Options | Custom-configured for specific process piping requirements; tri-clamp and specialty fittings available (1.5"–12") | | Compliance & Certifications | FDA 21 CFR 177.2600 & 1550, 3-A approved materials, USDA compliant, ISO9001 Approved; complete component passivation available |
Artesian's product line includes tri-clamp screen gaskets in VITON/FKM, PTFE/Teflon, and BUNA-N materials, offering filtration ratings from 5 to 100 microns. All wetted materials meet USP Class VI cytotoxicity testing and operate within -40°F to +250°F.

Each of these vendors addresses a distinct operational need — from validated single-use bioprocessing (Pall, Sartorius) to hygienic housing design (Parker), high-throughput gas and liquid filtration (Donaldson), and fully custom process engineering (Artesian Systems). The right fit depends on your process scale, regulatory submission requirements, and whether your application demands off-the-shelf validation packages or a purpose-built system.
Key Features to Look for in Pharmaceutical Inline Filters
Selecting the right inline filter comes down to four core criteria: micron rating, sanitary design compliance, material certifications, and integrity testability. Each one directly affects your validation strategy and regulatory standing.
Micron Rating and Filtration Mechanism
The required micron rating depends on the application:
- Sterilizing filtration: 0.2 µm or smaller (0.22 µm and 0.2 µm are considered interchangeable)
- Clarification or particle removal: 1–10 µm depth or surface filtration
- Bioburden reduction: 0.45–1.0 µm membrane or nominal depth filters
Filtration mechanisms include:
- Membrane filters: Provide absolute retention at defined pore size; used for final sterilizing filtration (materials: PES, cellulose acetate, PTFE, modified nylon)
- Depth filters: Use graduated porosity media for clarification and prefiltration of high-particulate loads; not validated as sterilizing filters
- Sintered metal: Reusable, high-temperature stable filters for gas/vent applications

Important: The FDA notes that Acholeplasma laidlawii can penetrate 0.2 µm filters but is retained by 0.1 µm filters. Validation with actual process fluid is critical, not just water challenge.
Sanitary Design Compliance
Filters for pharmaceutical piping must conform to hygienic design standards:
- ASME BPE: Surface finish classes SF1 (Ra < 20 µin) to SF4 (Ra < 15 µin with electropolishing); dead-leg L/D ratio of 2:1 or better (1.5:1 is now the preferred standard in biotech)
- 3-A Sanitary Standards: Clamp connections rather than threaded; surfaces free from pits and crevices; mechanically cleanable and easily dismantled
- ISO 14159: Supplemental hygienic design principles for process equipment
Non-compliant designs create harboring points for biofilm. Systems should be sloped to be self-draining (minimum 1/8 inch per foot or 1%).
Material Compatibility and Regulatory Certifications
Wetted materials must be:
- FDA 21 CFR compliant: 21 CFR 177.1550 covers perfluorocarbon resins (PTFE, FEP, PFA); 21 CFR 177.2600 covers rubber articles (silicone, EPDM gaskets) for repeated use
- USP Class VI tested: Biological reactivity testing for plastic filter components
- Free from extractable compounds that could adulterate the product stream
Standard wetted materials include 316L stainless steel (minimum), PTFE, and select PVDF components for pharmaceutical contact.
Integrity Testability and Validation Support
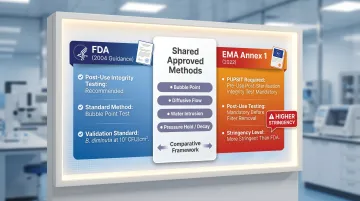
Sterilizing-grade inline filters must be integrity-testable in-line without removal:
- FDA (2004): Recommends post-use integrity testing routinely; bubble point testing is the standard approach
- EMA Annex 1 (2022): Requires pre-use post-sterilisation integrity testing (PUPSIT) and post-use testing prior to filter removal — a more stringent requirement than FDA
Approved test methods include bubble point, diffusive flow, water intrusion, or pressure hold/decay.
Vendors should supply validation documentation including:
- Extractables/leachables data
- Bacterial challenge test data (validated with B. diminuta at 10^7 CFU/cm² minimum)
- Drug master files (DMFs) where required by FDA or EMA
How We Chose the Best Inline Filtration Solutions
Evaluation Approach
Solutions were assessed on filtration technology breadth, sanitary construction standards, regulatory compliance documentation, and capacity to support pharmaceutical process engineers — from standard catalog products to custom-engineered assemblies.
Those criteria exist for a reason. A common mistake is selecting on price or brand name alone, without confirming that the filter's wetted materials, micron rating, and hygienic design match the specific process fluid and cleaning protocol.
Business-Outcome Factors
Three factors shaped the final rankings:
- Certifications (3-A, ASME BPE, FDA 21 CFR, cGMP documentation) reduce validation burden and regulatory risk
- CIP/SIP compatibility, integrity testability, and dead-leg-free geometry lower contamination risk and extend uninterrupted production runs
- For custom or specialty applications, support depth determines how quickly issues get resolved and how well the system scales as processes change
Conclusion
Choosing the right inline filtration solution for pharmaceutical process piping requires matching the filter's mechanism, micron rating, material certifications, and hygienic design to the specific demands of each process stage, fluid type, and cleaning protocol in your facility.
When evaluating vendors, look beyond product range. Engineering support, compliance documentation, and scalability matter — especially for non-standard or high-purity applications where catalog solutions won't cover your requirements.
If your pharmaceutical process piping requires a custom-engineered inline filtration solution built to exact sanitary specifications, specialty alloy construction, or unique process geometry, Artesian Systems provides complete process pipe engineering services and inline filtration design backed by FDA 21 CFR, 3-A, and ISO9001 credentials.
Reach out to discuss your specific application:
- Email: artesiansystems@gmail.com
- Phone: +1 (801) 318-4079
Frequently Asked Questions
What type of filtration is used in pharmaceutical engineering?
Pharmaceutical engineering uses membrane filtration (sterilizing-grade at 0.2 µm), depth filtration (clarification and prefiltration), ultrafiltration (protein separation), and inline process filters. The type selected depends on the manufacturing stage and the nature of the fluid being processed.
What is inline filtration in pharmaceutical manufacturing?
Inline filtration refers to filter assemblies integrated directly into the process piping run, allowing continuous removal of particulates, bioburden, or contaminants without diverting flow. These filters must meet sanitary design standards (ASME BPE, 3-A) for pharmaceutical use.
What certifications should inline filters have for pharmaceutical process piping?
Inline filters require FDA 21 CFR compliant wetted materials and USP Class VI certification at minimum, with 3-A Sanitary Standards approval or ASME BPE compliance strongly preferred. Sterile applications additionally require bacterial challenge data and integrity test documentation.
Do RO filters remove pharmaceuticals?
Reverse osmosis membranes reject many pharmaceutical compounds through size exclusion and charge effects, but removal efficiency varies by compound. In pharmaceutical manufacturing, RO is primarily used to produce purified water and WFI — not to filter product streams directly.
Which is better, F7 or M5 filter?
F7 and M5 are HVAC air filter classifications (EN779/ISO 16890) for facility air handling — not process piping. F7 offers higher particulate efficiency than M5, but neither rating applies to inline pharmaceutical filtration, where micron ratings, bacterial retention, and ASME BPE/3-A sanitary standards are the relevant benchmarks.
What is the difference between depth filtration and membrane filtration in pharmaceutical piping?
Depth filters trap particles throughout the filter matrix and are used for clarification and prefiltration of high-particulate loads. Membrane filters provide absolute retention at a defined pore size (such as 0.2 µm for sterilization) and are used for bioburden reduction or sterile filtration. In pharmaceutical process piping, both are often used in series.


