
Introduction
Material selection in process industries — where chemicals, high temperatures, and regulatory standards intersect — determines whether systems run reliably or fail catastrophically. A seal that swells in butter oil during a CIP cycle, an O-ring that hardens after repeated steam sterilization, or a gasket that leaches plasticizers into a pharmaceutical batch: these aren't hypothetical scenarios.
They're real failure modes that trigger batch recalls, regulatory citations, and six-figure downtime events.
FKM fluoroelastomer rubber is often reduced to a line item on a specification sheet. Its real-world value shows up in fewer maintenance cycles, audits passed without material deviations, and lower total cost of ownership driven by what doesn't fail. This guide covers five concrete benefits of FKM across food processing, pharmaceutical, chemical, and petroleum applications — grounded in how it performs when process conditions become aggressive.
TL;DR
- FKM withstands aggressive acids, petroleum fuels, and CIP chemicals that cause EPDM and NBR to swell or degrade
- Operates continuously at temperatures up to 204°C, maintaining sealing force through SIP cycles that exceed NBR's 125°C limit
- FDA 21CFR177.2600/1550 and 3A-certified grades support compliant sealing in food, beverage, and pharmaceutical applications
- FKM resists compression set and ozone degradation, extending service life and cutting spare parts inventory costs
- One FKM spec can cover acids, caustics, and solvents — reducing SKU count in multi-fluid processing facilities
What Is FKM Fluoroelastomer Rubber?
FKM is a class of synthetic fluorinated rubber standardized under ASTM D1418, built from fluorine-rich polymer chains (62–70% fluorine content by weight) that deliver extreme resistance to heat, chemicals, and aging.
The designation covers vinylidene fluoride (VDF) copolymers, primarily VDF-HFP, VDF-HFP-TFE, and VDF-TFE-PMVE formulations, each engineered for environments where standard elastomers degrade within hours.
In industrial settings, FKM appears as sealing and containment components across food, pharma, chemical, and petroleum sectors, including:
- O-rings and gaskets in process piping and reactors
- Seals and diaphragms in pumps and filtration systems
- Tubing in high-purity fluid handling lines
Artesian Systems sources and manufactures tri-clamp gaskets using FDA 21CFR177.2600/1550, USDA, and 3A-compliant FKM formulations for sanitary process systems handling everything from dairy CIP cycles to pharmaceutical USP water lines.
For any process line where seal failure means product contamination, a failed compliance audit, or premature replacement, FKM is the practical engineering choice — not an over-specified one.
5 Key Benefits of FKM Fluoroelastomer Rubber
The following benefits focus on operational outcomes measured in failure rates avoided, audit findings prevented, and total cost reduced — not theoretical material properties disconnected from real process conditions.
Benefit 1: Exceptional Chemical Resistance
FKM's fluorine-carbon backbone makes it one of the most chemically inert elastomers available, maintaining structural integrity when exposed to concentrated acids, petroleum hydrocarbons, chlorinated solvents, and aromatic compounds that dissolve or swell competing materials.
Real-world relevance: Immersion testing by Precision Polymer Engineering exposed FKM, EPDM, and silicone gaskets to butter oil for 22 hours at 70°C — conditions simulating dairy processing with fat-based product contact. Results:
- FKM: 0% volume change, 0 Shore A hardness loss
- EPDM: 14.1% volume swell, -11 Shore A hardness loss
- Silicone: 0.1% volume change, 0 Shore A hardness loss
EPDM's double-digit swelling in fatty media renders it unsuitable for sealing applications in dairy, food oil processing, or any line handling lipid-rich products. FKM maintains dimensional stability where EPDM seals would lose compression force and leak.

Chemical degradation of seals is a leading cause of unplanned shutdowns and contamination events in process industries. When an NBR gasket swells in a CIP solvent or an EPDM seal leaches plasticizers into a pharmaceutical batch, the downstream cost isn't just the seal — it's the lost product, the batch rework, and in regulated industries, the recall.
Industry data shows the average food recall costs approximately $10 million, with over 15.5 million pounds of food recalled in a single year due to foreign material contamination.
KPIs impacted: Seal service life in chemical environments, CIP/SIP cycle frequency without component replacement, unplanned downtime incidents linked to seal failure, contamination event rates.
When this advantage matters most: Facilities running aggressive alkaline or acidic cleaning protocols, process lines handling petroleum derivatives or aromatic hydrocarbons, food processing operations with high-fat product contact, and any system where solvent or fuel exposure occurs routinely.
Benefit 2: High-Temperature Performance
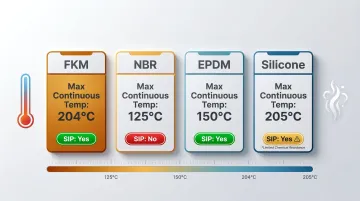
Chemical resistance only matters if the seal survives the heat. FKM maintains elastic properties from -40°C to over 200°C continuous service — Chemours rates Viton FKM at +204°C (+400°F) continuous, with intermittent exposure to 232°C. Compare this to alternatives:
| Material | Max Continuous Temp | Suitable for SIP? |
|---|---|---|
| FKM | 204°C (400°F) | Yes |
| NBR | 125°C (250°F) | No |
| EPDM | 150°C (300°F) | Yes |
| Silicone | 205°C (400°F) | Yes (limited chemical resistance) |
Steam-in-place (SIP) sterilization cycles operate at 121-135°C for sustained periods. NBR's 125°C ceiling falls within the SIP operating range — meaning NBR seals experience thermal stress at or above their maximum rated temperature during every sterilization cycle. PPE technical documentation notes that SIP temperatures "can reach up to 135°C, which is beyond the sealing capability of some elastomer material types."
FKM's 204°C rating provides substantial thermal margin, enabling the material to survive thousands of SIP cycles without the compression set (permanent deformation) that causes standard elastomers to lose sealing force. ASTM D395 Method B compression set testing shows FKM at 10-30% compression set after 70 hours at 200°C — maintaining elastic recovery where NBR would take permanent set and fail.
Thermal degradation of seals is often invisible until failure occurs. A gasket that looks intact externally may have lost 40% of its elastic memory internally, no longer generating sufficient sealing force. FKM's stable performance under heat removes this common failure mode from high-temperature process systems.
KPIs impacted: Seal replacement frequency during thermal cycling, compression set percentage over time, SIP/steam sterilization cycle tolerance, mean time between seal failures in heated systems.
When this advantage matters most: Sterilization-intensive pharmaceutical and food processing operations, high-temperature petroleum refining and chemical reactor applications, pasteurization systems, and any process line undergoing regular SIP or autoclave-level heat exposure.
Benefit 3: Regulatory Compliance for Food, Pharma, and Sanitary Applications
Temperature and chemical performance get FKM on the shortlist. Compliance keeps it there. FKM formulations meeting FDA 21 CFR 177.2600 and 3-A Sanitary Standard 18-03 are specifically engineered for direct contact with food, beverages, pharmaceuticals, and potable water. This is not a default property of all FKM grades — it's a function of validated polymer formulations and extraction limits.
FDA 21 CFR 177.2600 specifies:
- Approved polymer types: VDF-HFP copolymers (minimum molecular weight 70,000) and VDF-HFP-TFE copolymers (minimum molecular weight 100,000)
- Extraction limits for aqueous food contact: maximum 20 mg/sq in total extractives (first 7 hours)
- Extraction limits for fatty food contact: maximum 175 mg/sq in (first 7 hours)
3-A Sanitary Standard 18-03 classifies FKM as Class I — the highest material tier alongside FFKM and silicone — based on passing CIP chemical compatibility testing at 82°C, milkfat absorption tests, and physical property retention requirements. EPDM and NBR are classified as Class II.
Compliance-grade FKM enables operations to withstand FDA inspections, 3-A certification audits, and USDA facility approvals without material substitutions or compliance waivers. USDA guidelines require formal documentation of CFR compliance "on company letterhead, with material trade name and signature of certifying official" — shifting compliance from an assumed material property to an active documentation requirement.
Using non-compliant elastomers in food or pharma lines creates direct regulatory liability. A seal material that doesn't meet 21 CFR 177.2600 can trigger FDA Form 483 observations or void facility certifications.
Compliance-grade FKM with verified certifications removes that liability and simplifies the material traceability chain during audits.

KPIs impacted: Audit finding frequency related to material compliance, number of material deviations in batch production records, regulatory inspection outcomes, documentation time for material traceability during audits.
When this advantage matters most: FDA-regulated pharmaceutical manufacturing, 3-A-certified dairy and food processing, USDA-inspected meat and poultry facilities, and any process where direct product-contact materials must carry validated compliance documentation.
Benefit 4: Superior Durability and Reduced Maintenance Intervals
Compliance addresses regulatory risk. Durability addresses operating cost. FKM's dense molecular structure resists compression set, ozone exposure, UV degradation, and oxidative aging — meaning seals maintain their geometry and elastic recovery longer than silicone or general-purpose rubbers in comparable service.
Ozone resistance testing at 150 ppm for 2 weeks rates FKM as "Outstanding" alongside EPDM, while NBR is rated "Not Recommended." Weather and oxidation resistance follow the same pattern: FKM and EPDM excel, while NBR degrades rapidly in outdoor or UV-exposed installations.
The operational impact: longer-lasting seals translate to fewer planned and unplanned replacement events, reduced spare parts inventory costs, and lower cumulative labor spend on system teardown and reassembly. In continuous-process operations where a shutdown costs $10,000-50,000 per hour, eliminating even one unplanned seal replacement event per quarter delivers measurable ROI.
Industry guidance recommends seal inspection every 6-12 months in food processing environments — but those intervals assume the seal material can survive service conditions between inspections. FKM's resistance to compression set and environmental degradation makes those intervals achievable; NBR or silicone in aggressive chemical or thermal service may not reach the first inspection without degradation.
KPIs impacted: Mean time between seal replacements (MTBR), planned maintenance labor hours per year, spare parts inventory carrying costs, overall equipment effectiveness (OEE) impact from seal-related downtime.
When this advantage matters most: Continuous-process operations where shutdowns carry high opportunity costs, high-cycle systems (pumps, valves, filtration units with frequent pressure/temperature swings), and facilities in remote locations where component replacement requires logistical coordination.
Benefit 5: Broad Fluid and Media Compatibility
Durability extends FKM's value within a single process line. Broad fluid compatibility extends it across entire facilities.
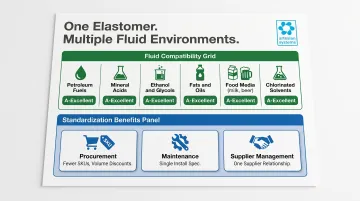
FKM chemical compatibility data confirms A-Excellent ratings across a wide range of process fluids:
- Petroleum fuels, diesel oil, hexane, biodiesel
- Concentrated mineral acids: HCl 37%, HNO₃, H₂SO₄ 75-100%, H₃PO₄
- Ethanol, glycols, fats, oils, and greases
- Milk, beer, and other food-grade media
This breadth of compatibility allows facilities running multiple fluid types to standardize on FKM rather than stocking EPDM for one line, NBR for another, and silicone for a third. Standardization simplifies operations across the board:
- Procurement: fewer SKUs and higher volume discounts
- Maintenance: one set of installation specs and torque values
- Supplier management: a single material supplier relationship
FKM's known incompatibilities — ketones (acetone, MEK), low-molecular-weight esters, strong alkalis above 20% concentration, and amines — are thoroughly characterized. Engineers can select FKM confidently for roughly 80% of industrial fluid environments and specify alternatives only where the chemistry requires it.
When this advantage matters most: Multi-product facilities producing different goods on shared equipment, hemp and cannabis extraction operations handling both hydrocarbon solvents and ethanol, petroleum and chemical plants managing diverse fluid streams within a single process unit, and any operation seeking to reduce elastomer inventory complexity.
What Happens When You Use the Wrong Elastomer
Elastomer incompatibility manifests in measurable, costly ways:
Seal swelling or shrinkage: An EPDM gasket exposed to petroleum derivatives swells beyond its groove dimensions, losing compression and creating leak paths. An NBR seal in biodiesel shrinks, opening gaps that allow product contamination.
Extractables contaminating product streams: Elastomers contain curing agents (bisphenol AF, organophosphonium salts), plasticizers, and processing aids. When seals degrade in aggressive media, these compounds leach into the process stream, causing off-flavors in food products, failing USP purity requirements in pharmaceuticals, or introducing endocrine disruptors into consumer goods.
Compression set causing loss of sealing force: A silicone gasket subjected to repeated SIP cycles takes permanent set — it no longer rebounds to fill the sealing surface. The result is a slow leak that may not appear during initial testing but emerges weeks later during production.
Accelerated degradation under thermal or chemical cycling: PPE technical documentation states that acidic CIP washes can cause seal failure "almost immediately" with incompatible materials. The time between installation and failure compresses from months to days.
In regulated industries, a single contamination event traced to an incompatible seal can trigger full batch recalls, regulatory inspections, or loss of facility certifications. The average food recall costs $10 million — against a correct seal that typically runs $5 to $50. That cost gap is what makes material selection a risk management decision, not just a purchasing one.

Elastomers also fail in ways that aren't immediately obvious. A seal can appear intact externally while leaching plasticizers internally or losing 50% of its elastic memory without visible surface cracks. Without proper material specifications and traceability documentation, failures surface only after the damage is done.
How to Get the Most from FKM Rubber in Process Systems
FKM performs best when correctly specified from the start. Material grade selection should be based on:
- Specific chemical environment — not all FKM grades resist all chemicals equally. Higher fluorine content (68-70%) improves fuel and solvent resistance; lower fluorine content (65-66%) improves low-temperature flexibility.
- Temperature range — continuous exposure above 200°C or below -20°C requires specialty FKM formulations.
- Regulatory requirements — verify FDA, 3A, or USDA compliance at the component level, not the polymer class level.
Not all FKM carries food-contact compliance. FDA does not "approve" elastomer compounds — manufacturers must perform extraction testing per 21 CFR 177.2600 and issue certificates of compliance based on results.
Procurement teams must request and verify these certificates. Assuming compliance based on "FKM" as a material class won't hold up in an audit.
Verified compliance covers the material side. Correct installation determines how well that material performs in service:
- Squeeze ratios: 15-30% for static seals, 8-20% for dynamic seals
- Surface finish: 0.4-1.6 Ra for contact surfaces, per Parker O-Ring Handbook
- Torque procedures: Follow manufacturer specifications to avoid over-compression
Documented maintenance schedules should include inspection intervals every 6-12 months, checking for compression set, surface cracking, and hardening. Catching these signs early extends component life and prevents unplanned failures.
Working with suppliers who provide material certifications, traceability documentation, and application-specific guidance reduces specification risk and audit exposure. Artesian Systems manufactures tri-clamp gaskets with FDA 21 CFR 177.2600/1550, USDA, and 3A compliance verified at the component level — with the certification documentation required for regulated facility audits available at the time of purchase.
Conclusion
FKM fluoroelastomer rubber's advantages — chemical resistance, thermal stability, regulatory compliance, durability, and fluid versatility — compound to reduce operational risk across the board. Chemical resistance prevents contamination events. Thermal stability extends service life through SIP cycles. Compliance certification simplifies audits. Durability reduces maintenance frequency. Fluid compatibility enables material standardization.
The true value of FKM shows up over time — in shutdowns that don't happen, audits that pass clean, and seals that reach their second year instead of failing in the first quarter. That reliability comes from a material engineered, tested, and documented specifically for the conditions it faces.
Material selection is an engineering and compliance decision — one worth revisiting as process conditions and regulatory standards shift. The real cost comparison isn't FKM versus a cheaper elastomer at the point of purchase. It's FKM versus the downtime, contamination risk, and audit exposure that come when the wrong seal fails mid-process.
Frequently Asked Questions
What is FKM fluoroelastomer?
FKM is a synthetic rubber class defined under ASTM D1418, distinguished by high fluorine content (62-70% by weight) that delivers exceptional resistance to heat, chemicals, and aging. It's widely used in seals, O-rings, and gaskets across food, pharmaceutical, petroleum, and chemical processing industries.
Is FKM fluoroelastomer the same as Viton, Teflon, or silicone?
Viton is DuPont/Chemours' brand name for FKM — both refer to the same fluoroelastomer material class. Teflon (PTFE) is a distinct fluoropolymer that is rigid and non-elastomeric. Silicone is a separate synthetic rubber with lower chemical and high-temperature resistance than FKM.
Is FKM fluoroelastomer rubber safe for food and pharmaceutical contact?
FKM formulations meeting FDA 21CFR177.2600/1550, 3A Sanitary Standards, and USDA criteria are validated for direct contact with food, beverages, and pharmaceutical products. Not all FKM grades carry these certifications, so compliance must be verified at the component level through supplier documentation before use.
How can I verify if FKM components are PFAS-free?
FKM is technically classified as PFAS due to fully fluorinated carbon atoms, but is categorized as a "Polymer of Low Concern" because of its high molecular weight and chemical stability. To verify compliance, request the following from your supplier:
- Material Safety Data Sheets (MSDS)
- Certificates of Analysis
- REACH/RoHS compliance statements
- PFAS-free processing aid declarations
Is FKM better than silicone for seals and gaskets in process applications?
In food, pharma, and chemical applications, FKM outperforms silicone in resistance to acids, oils, and hydrocarbons, and holds up better under sustained compression load. Silicone suits lower-demand applications, but is not a substitute where chemical aggression or regulatory compliance is a factor.


