
Introduction
In regulated industries like food processing, pharmaceuticals, and hemp extraction, a single wrong gasket material choice can trigger contamination events, failed audits, or unplanned downtime. Yet gaskets are routinely selected based on availability or cost rather than engineered compatibility with the actual process conditions they'll face.
When a gasket fails prematurely, the consequences extend far beyond the part itself. Contamination can ruin product batches worth thousands of dollars. Unplanned downtime in chemical processing plants can cost $500,000 per day or more. Failed audits jeopardize certifications that entire operations depend on.
This guide covers the most common sanitary gasket materials—their chemical compatibility, temperature limits, and compliance credentials—along with a structured framework for matching material to application. Whether you're running a CIP-heavy dairy line or a solvent-based extraction process, you'll find a clear path to the right choice.
TL;DR
- Material choice directly determines chemical resistance, thermal performance, and regulatory compliance
- Five primary types covered: EPDM, FKM (Viton), Platinum Cured Silicone, Nitrile/Buna-N, and PTFE
- Match each material against your process chemicals, temperature range, pressure, and CIP/SIP requirements
- FDA 21 CFR, 3-A, and USDA compliance requirements vary by material — verify before specifying
- Color dot codes molded into gasket faces allow quick field identification without pulling spec sheets
Common Sanitary Gasket Materials: Properties, Applications, and Color Codes
Each material has a distinct chemical, thermal, and regulatory profile. The right choice depends on process fluid, cleaning regime, and purity requirements. Most gaskets appear black or white regardless of material, which is why color dot codes and documentation matter.
EPDM (Ethylene Propylene Diene Monomer)
EPDM ranks among the most widely used sanitary gasket elastomers due to excellent resistance to water, steam, alcohols, ketones, and mild acids/alkalis. It operates continuously from -30°F to 300°F (-34°C to 149°C), making it suitable for low-pressure steam CIP applications.
The curing method determines purity level—an important distinction for pharmaceutical buyers:
- Sulfur-cured (one green dot): Standard formulation
- Peroxide-cured (three green dots): Lower extractables, preferred for pharmaceutical applications
EPDM is incompatible with oils and hydrocarbon-based products — exposure causes swelling and seal failure. This rules it out for cannabis extraction systems using butane or propane, and for food processing lines handling oils or fats.
Compliance: FDA 21 CFR 177.2600, USP Class VI, 3A Sanitary Standards, USDA
FKM / Viton (Fluoroelastomer)
FKM is the preferred choice for CIP and SIP systems due to broad resistance to acids, alkalis, alcohols, solvents, and oils. It operates continuously from -30°F to 400°F (-34°C to 204°C), with short-term exposure capability up to 600°F (315°C).
Identification: One yellow dot + one white dot
Three FKM formulations are available, each targeting different process demands:
- Viton A: Most cost-effective; excellent oil and steam resistance
- Viton GF: High-fluorine, peroxide-cured; enhanced caustic CIP resistance
- Viton ETP: Broadest chemical resistance of the three; lower-cost alternative to FFKM
Performance degrades in hot, concentrated caustic solutions and certain ketones. While FKM excels in acid service, it's rated only "Acceptable" for continuous steam — not ideal for intensive SIP protocols.
Compliance: FDA 21 CFR 177.2600, USP Class VI, 3A Sanitary Standards, USDA
Platinum Cured Silicone
Platinum cured silicone is the material of choice for ultra-high purity pharmaceutical and biotech applications. Its addition cure process produces no byproducts, resulting in extremely low extractables and leachables. Operating range: -40°F to 450°F (-40°C to 232°C).
Identification: No color dot (typically translucent or white)
The trade-off is cost. Platinum cured silicone is the most expensive standard option, making it less common in general food processing but strongly preferred for injectable drug manufacturing and medical device applications.
Unlike peroxide-cured silicone (one pink dot), which produces volatile organic acids during curing, platinum-cured formulations meet USP Class VI requirements and offer superior purity for sensitive processes.
Compliance: FDA 21 CFR 177.2600, USP Class VI, 3A Sanitary Standards, USDA
Nitrile / Buna-N (NBR)
Buna-N is the most economical sanitary gasket material, offering good compatibility with moderate chemicals, mineral oils, and fats. Continuous service temperature: -30°F to 200°F (-34°C to 93°C).
Identification: One red dot
Critical limitations to know before specifying:
- Does NOT pass USP Class VI certification
- Not recommended for CIP applications
- Rated "Poor" for pure water service
- Rated "Failure" for steam service
Suitable only for ambient and potable water lines, or lower-purity general process applications where budget constraints outweigh performance requirements.
Compliance: FDA 21 CFR 177.2600, 3A Sanitary Standards (food grade only)
PTFE (Polytetrafluoroethylene)
PTFE is a plastic, not a rubber — meaning it has minimal compression memory and limited "squish factor." Operating range: -100°F to 500°F (-73°C to 260°C).
This creates sealing challenges, especially in larger sizes, but its chemical inertness is unmatched across virtually all process fluids and cleaning agents. PTFE is rated "Excellent" for steam, pure water, and all CIP/SIP media.
PTFE suffers from cold flow — permanent deformation under sustained compression. Because of this, the 150 PSI service rating for sanitary clamps does not apply to connections 1" and larger with PTFE gaskets.
PTFE envelope gaskets address this directly. They combine a PTFE outer shell with an elastomeric core (typically FKM or EPDM). This unitized design (a single assembled gasket unit) provides PTFE's chemical resistance with the sealing memory of an elastomer, reducing cold flow and improving performance in misaligned ferrules.
Identification: No color dot (white/translucent)
Compliance: FDA 21 CFR 177.1550, USP Class VI, 3A Sanitary Standards, USDA
Specialty and High-Performance Materials
Kalrez (FFKM) is a perfluoroelastomer offering near-universal chemical inertness and elevated temperature resistance up to 428°F (220°C). It's designed for the most aggressive CIP/SIP/WFI applications in pharmaceutical and biotech environments.
Chemical resistance rivals PTFE while maintaining elastomeric sealing performance. Kalrez meets USP Class VI requirements when tested at 121°C.
Kalrez costs approximately 5–20x more than standard FKM. Reserve it for processes where FKM or PTFE fall short on chemical compatibility or temperature requirements.
Other engineered options exist for edge cases where standard materials fall short:
- Sanifluor: Mid-tier fluoroelastomer with improved caustic resistance vs. standard FKM
- Custom elastomer blends: Designer compounds formulated for specific fluid/temperature combinations
- Proprietary composites: Available from specialty manufacturers for highly unusual service conditions
Select these with engineering guidance — compatibility data from the fluid manufacturer is essential before specifying.
Critical Factors for Sanitary Gasket Material Selection
Selecting gasket material is not just a chemical compatibility lookup. It requires connecting process operating conditions, cleaning protocols, regulatory requirements, and total cost of ownership to ensure the gasket performs reliably without compromising product purity.
Chemical Compatibility with Process and Cleaning Fluids
This is the primary filter. The gasket must be inert to both the product being processed and the CIP/SIP agents used.
Common CIP chemicals include:
- Caustic wash (sodium hydroxide): 0.5%-2.0% concentration, 140°F-185°F
- Acid wash (nitric acid): 0.5% concentration, 130°F-150°F
- Sanitizers: sodium hypochlorite, chlorine dioxide, peracetic acid
- Sterilization (SIP): high-pressure steam at approximately 250°F for 30 minutes
A material that handles the product but degrades under cleaning agents will fail prematurely. EPDM handles caustic service well but struggles with acids. FKM holds up under acid wash yet cannot handle continuous steam. When breadth of compatibility matters most, PTFE and FFKM cover the widest range of both process fluids and cleaning agents.
| Chemical | EPDM | Buna-N | Silicone | FKM | PTFE |
|---|---|---|---|---|---|
| Sodium Hydroxide | Excellent | Good | Good | Good | Excellent |
| Nitric Acid | Good | Not Recommended | Good | Excellent | Excellent |
| Phosphoric Acid | Excellent | Good | Good | Excellent | Excellent |
| Sodium Hypochlorite | Good | Good | Good | Excellent | Excellent |
| Steam (to 400°F) | Limited | Not Recommended | Not Recommended | Not Recommended | Limited |

Operating Temperature Range
Gaskets must maintain seal integrity across the full thermal cycle—from cold storage or water rinse through hot processing or sterilization. Exceeding rated temperature causes compression set, cracking, or extrusion.
Temperature thresholds by material:
- EPDM: -30°F to 300°F continuous; acceptable for low-pressure steam
- FKM: -30°F to 400°F continuous; short-term to 600°F
- Platinum Silicone: -40°F to 450°F continuous
- Buna-N: -30°F to 200°F continuous (lowest thermal capability)
- PTFE: -100°F to 500°F continuous (widest range)
- Kalrez (FFKM): Up to 428°F continuous
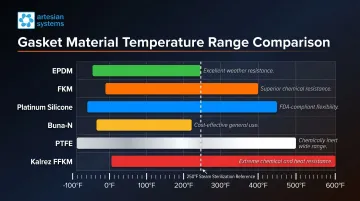
If your process alternates between 40°F cold water rinse and 250°F steam sanitization, Buna-N fails immediately. EPDM works but sits at the edge of its range. FKM or platinum silicone provides margin.
System Pressure and Mechanical Load
Internal operating pressure affects both gasket compression and the risk of extrusion past clamp ferrules.
PTFE requires careful torque management due to cold flow. Standard sanitary clamps rated at 150 PSI do not maintain that rating for connections 1" and larger when using PTFE gaskets. Fractional sizes (½" and ¾") are exempt due to recessed, fully contained design.
Elastomers must have sufficient durometer (hardness rating) to resist extrusion at high pressures without over-compressing and cracking. PTFE envelope gaskets with elastomeric cores solve this by combining PTFE's chemical resistance with the mechanical resilience of EPDM or FKM.
Regulatory and Compliance Requirements by Industry
The target industry drives which certifications are non-negotiable:
- Pharmaceutical and biotech: USP Class VI required; often 3A Sanitary Standards
- Food and beverage: FDA 21 CFR 177.2600 minimum
- Cannabis/hemp extraction: Materials compatible with hydrocarbon solvents (butane, propane, ethanol)
| Standard | Applies To | Key Requirement | Materials That Qualify |
|---|---|---|---|
| FDA 21 CFR 177.2600 | Food-contact rubber | Extractives testing; no harmful leachables | EPDM, FKM, Silicone, NBR |
| FDA 21 CFR 177.1550 | Food-contact PTFE/fluoropolymers | Temperature limits; extractable fluoride testing | PTFE, FFKM |
| USP Class VI | Pharma/biotech contact materials | Systemic injection, intracutaneous & implantation tests at 121°C | Platinum-cured silicone, FFKM |
| 3A Sanitary Standards | Dairy and food processing | Acid, alkaline, chlorine, and heat resistance; Class I to 300°F, Class II to 250°F | EPDM, FKM, PTFE (material-dependent) |
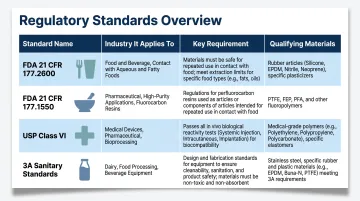
Mismatched certifications are caught in audits — and correcting them mid-production is far more disruptive than getting material selection right upfront.
Total Cost and Replacement Frequency
Upfront material cost is misleading. A premium gasket material may cost more per unit but outlast lower-cost options 3-5x in aggressive chemical environments.
Downtime in chemical processing plants costs an estimated $500,000 per day or more. Premature gasket failure drives replacement labor, contamination risk, and production loss that far exceeds material cost differential.
Lifecycle considerations:
- EPDM gaskets that bond to stainless flanges can damage ferrules and inflate maintenance hours over time
- FKM formulations validated to 500 clean steam cycles cut replacement frequency significantly
- Kalrez costs more per unit, but in aggressive chemical service its seal life typically justifies the premium
Total cost of ownership = (material cost + installation labor + replacement frequency) × (downtime risk + contamination risk).
Regulatory Compliance and Certification Standards for Sanitary Gaskets
Three primary compliance benchmarks govern sanitary gasket materials:
FDA 21 CFR 177.2600 applies to rubber articles — EPDM, FKM, silicone, and Buna-N — intended for repeated food contact. Aqueous food contact requires meeting water extractives limits; fatty food contact adds chloroform-soluble extractives requirements.
FDA 21 CFR 177.1550 governs PTFE, FEP, and PFA resins for food-contact use. These materials may be used as articles or components contacting food, subject to temperature limits and extractable fluoride testing.
USP Class VI (USP <87> and <88>) is the most rigorous biocompatibility standard, requiring three in vivo tests: systemic injection (mice), intracutaneous (rabbits), and implantation (rabbits, 120+ hours). Extractions run at 121°C (autoclaved for 60 minutes), the highest extraction temperature, making USP Class VI the gold standard for biocompatibility testing.
3A Sanitary Standards 18-03 covers multiple-use rubber and rubber-like materials for product contact surfaces in dairy equipment. Four temperature classes define service limits:
- Class I — up to 300°F
- Class II — up to 250°F
- Class III — up to 120°F
- Class IV — up to 100°F
Materials must withstand accelerated use tests involving nitric acid, phosphoric acid, alkaline cleaners, and chlorine sanitizers.
For buyers who need to meet multiple standards simultaneously, Artesian Systems stocks materials compliant with FDA 21 CFR 177.2600, 177.1550, USP Class VI, 3A Sanitary, and USDA requirements — so compliance verification doesn't fall on you.
Color Dot Identification System
Color dot codes are molded into the gasket side edge, enabling rapid visual material identification during maintenance without documentation:
| Color Code | Material |
|---|---|
| One red dot | Buna-N / NBR |
| One green dot | Sulfur-cured EPDM |
| Three green dots | Peroxide-cured EPDM |
| One yellow + one white dot | FKM / Viton |
| One pink dot | Peroxide-cured silicone |
| No dot | Platinum-cured silicone or PTFE |

This system is consistent across manufacturers and helps maintenance teams verify correct material installation in the field — critical when multiple material types are used in the same facility.
How Artesian Systems Can Help
Artesian Systems is a specialized engineering and components provider with almost two decades of experience in petroleum, chemical, food, and pharmaceutical industries. The company sources and supplies sanitary gasket materials pre-compliant with FDA 21 CFR 177.2600, FDA 21 CFR 177.1550, USDA, and 3A Sanitary Standards.
Standard inventory includes:
- EPDM: Available as core material in PTFE/EPDM envelope gaskets, sizes 1.5"–12"
- FKM (Viton): Screen gaskets with 5, 20, and 100 micron filtration, sizes 1.5"–6"
- Nitrile/Buna-N: Screen gaskets with 20 and 100 micron filtration, sizes 1.5"–6"
- PTFE: Unitized envelope gaskets with EPDM core, sizes 1.5"–12"; also available with integrated stainless steel screens

Complete process pipe engineering services and custom system design mean gasket material selection happens in context — accounting for CIP protocols, pressure ratings, and cleaning chemical compatibility — not in isolation. This is the difference between a component that fits and one that performs reliably over the life of the system.
Key differentiators:
- ISO9001 approved quality management
- CNC machining for specialty alloys and composite materials, including custom PTFE components
- Section VIII Division 1 vessel and reactor building capabilities
- Direct engineering consultation across food and beverage, pharmaceutical, nutraceutical, cosmetics, hemp/cannabis extraction, and chemical processing
For replacement gaskets or full system design, contact Artesian Systems directly at artesiansystems@gmail.com or call +(801) 318-4079.
Conclusion
Gasket material selection is an engineering decision with compliance, safety, and operational consequences. The right material must align with process chemistry, temperature profile, cleaning regime, and regulatory framework specific to your industry.
No single material excels across all conditions. Each option carries real limitations:
- EPDM handles caustic cleaning well but degrades in oils and solvents
- FKM resists most acids but breaks down in hot caustic environments
- PTFE offers broad chemical compatibility but cold-flows under compression over time
- Platinum silicone meets ultra-high purity requirements but carries a higher cost
Knowing these trade-offs before specifying a material prevents costly misapplication.
Material selection doesn't end at installation. Gaskets are consumable components, and the conditions they operate under rarely stay static. Process conditions shift, cleaning protocols change, and materials wear — making periodic review essential. A scheduled gasket audit catches degradation before it causes downtime or contamination.
For applications where the margin for error is tight — pharmaceutical processing, high-purity beverage lines, or extraction systems — working with engineers who understand both material science and process conditions is worth the investment. Artesian Systems provides process engineering consultation for exactly these decisions, from gasket specification through full system design.
Frequently Asked Questions
What is the most common sanitary gasket material for food and beverage processing?
EPDM is the most widely used due to its balance of chemical resistance, thermal performance, and cost-effectiveness. It's FDA 21 CFR 177.2600 compliant and well-suited to water, steam, and mild acid/alkali service. Peroxide-cured EPDM (three green dots) is preferred for pharmaceutical applications requiring lower extractables.
What does FDA 21 CFR 177.2600 compliance mean for sanitary gaskets?
This FDA regulation governs rubber articles intended for repeated food contact. Compliant materials have been tested to confirm they won't leach harmful substances into food under defined use conditions. EPDM, FKM, silicone, and Buna-N gaskets all fall under this standard.
Can EPDM gaskets be used in CIP and SIP applications?
EPDM handles CIP service well — water, steam, and alkaline cleaners are all within its range. For SIP systems at sustained high steam temperatures, it falls short. FKM is the go-to for combined CIP/SIP service, rated for continuous use up to 400°F.
What is the difference between platinum-cured and peroxide-cured silicone gaskets?
Platinum-cured silicone uses an addition cure process with no byproducts, giving it lower extractables and USP Class VI compliance — the standard for pharmaceutical use. Peroxide-cured silicone releases volatile organic acids during curing, making it more economical but unsuitable where high purity is required.
How can I identify a sanitary gasket material without documentation?
Check the standardized color dot system on the gasket side edge. Red dot = Buna-N, one green dot = sulfur-cured EPDM, three green dots = peroxide-cured EPDM, yellow + white dot = FKM. Platinum-cured silicone and PTFE have no dots but are visually distinct (translucent white).
Which sanitary gasket material is best for pharmaceutical or biotech applications?
Platinum-cured silicone suits applications requiring minimal extractables and ultra-high purity, while FKM is the preferred choice for CIP/SIP-intensive systems. For the most aggressive chemical environments where neither qualifies, Kalrez (FFKM) is the answer — at 5–20x the cost of standard FKM.


