
Introduction
In 2016, a leading peanut butter manufacturer faced two costly product recalls triggered by rubber fragments entering their process stream. Standard elastomeric gaskets degraded under the combined assault of high-viscosity product, abrasive nut particles, and aggressive vegetable oils. The financial impact extended beyond recall costs—production downtime, regulatory scrutiny, and brand damage compounded the total loss.
This isn't an isolated incident. Approximately 10% of all food recalls over the past 20 years stem from foreign material contamination, with gaskets and O-rings explicitly identified by USDA FSIS Directive 7310.5 as contamination sources in meat and poultry facilities.
This guide gives engineers, compliance managers, and procurement teams a practical framework for selecting food grade gaskets. It covers regulatory compliance (FDA 21 CFR, 3-A, NSF, USDA), material trade-offs across silicone, EPDM, nitrile, PTFE, and FFKM, and how to match gasket specifications to your process chemistry, temperature profile, and regulatory requirements.
TLDR:
- FDA compliance requires meeting extractive limits under 21 CFR 177.1210 or 177.2600 depending on application
- 3-A mandates Ra ≤ 32 μin surface finish and ≤ 1/32" offset for CIP joints—FDA alone isn't enough for dairy
- Silicone suits wide temperature ranges; EPDM excels in CIP; PTFE offers broadest chemical resistance
- FFKM costs 8–35x baseline elastomers—justified only for aggressive chemistry or high downtime risk
- Match certification to facility: 3-A for dairy/beverage, NSF 61 for potable water, USDA for meat/poultry
What Are Food Grade Gaskets and Why Do They Matter?
Food grade gaskets are sealing components manufactured from materials proven non-toxic, odorless, tasteless, and bacteria-resistant through standardized extractive testing. Unlike industrial gaskets that may contain fillers or plasticizers optimized for mechanical performance, food grade materials must demonstrate that harmful substances won't leach into food, beverages, or pharmaceutical products during processing.
That requirement isn't self-certified — it's governed by federal regulation.
Under FDA 21 CFR Part 177, these gaskets fall into two regulatory categories:
- Section 177.1210 governs closure-sealing gaskets for food containers (single-use or limited reuse)
- Section 177.2600 addresses rubber articles for repeated use in processing equipment — the piping gaskets, valve seals, and pump gaskets subjected to thousands of thermal and chemical cycles
How Gasket Failure Creates Contamination Risk
The functional consequences of gasket failure extend beyond simple leaks. When a gasket degrades in a food processing system, five distinct failure modes create direct contamination pathways:
| Failure Mode | Contamination Risk |
|---|---|
| Material degradation | Chemical leaching into product stream |
| Brittleness/fracture | Physical fragments shed into food |
| Microbial colonization | Bacteria harbored in gasket surface crevices |
| Compression set | Loss of seal integrity allowing foreign matter ingress |
| Extrusion damage | Frayed material entering product flow |

Regulatory Consequences of Gasket Failures
In 2022, FDA recorded 17 foreign material recalls out of 242 total recalls, with foreign material complaints representing 45% of all primary consumer complaints according to the FDA Consumer Complaint Monitoring System. USDA FSIS enforcement treats gasket contamination as a food safety event, not merely a maintenance issue — facilities face noncompliance records, product retention, mandatory recalls, and potential suspension of operations.
Artesian Systems manufactures tri-clamp gaskets (1.5" to 12") from materials compliant with FDA 21 CFR 177.2600 and 177.1550, with 3-A approved and USDA compliant options available. That means facilities in food, pharma, and beverage processing can meet both baseline FDA requirements and sector-specific standards from a single source.
Key Compliance Standards for Food Grade Gaskets
FDA 21 CFR 177.1210 vs. 177.2600: Which Applies?
FDA 21 CFR 177.1210 regulates closure-sealing gaskets for food containers—the seals on jar lids, bottle caps, and single-use packaging. Extractive limits are measured in parts per million based on water capacity: 50 ppm for water and alcohol extractives; 50-500 ppm for heptane depending on gasket type. Testing protocols vary by food storage temperature (250°F for heat-sterilized products down to 70°F for refrigerated storage).
FDA 21 CFR 177.2600 covers rubber articles intended for repeated food contact—processing gaskets in pipelines, vessels, and sanitary fittings. Extractive limits shift to area-based measurement: 20 mg/sq in for the first 7 hours of water extraction; 175 mg/sq in for n-hexane extraction. For fatty food contact, carbon black content cannot exceed 10% by weight.
Critical distinction: If you're sealing a processing pipeline that undergoes daily CIP cycles and handles product for years, 177.2600 applies. If you're sealing a container that ships to consumers, 177.1210 applies. When requesting compliance documentation, specify which regulation applies and reference the relevant food type from 21 CFR 176.170 Table I.
3-A Sanitary Standards: Design Requirements Beyond Material Compliance
3-A adds a hygienic design layer on top of FDA material approval. A gasket material can pass all FDA extractive tests yet fail 3-A if its geometry creates bacterial harborage or resists CIP cleaning.
- Surface finish: Ra ≤ 32 μin (0.8 μm), typically achieved by No. 4 finish stainless steel
- CIP joint geometry: Flat gaskets must be "substantially flush" with an offset ≤ 1/32 inch. If gasket thickness is less than 1/32", offset cannot exceed gasket thickness.
- Drainage design: Double-seal equipment requires visible leak detection paths open to atmosphere, located at the lowest point, with diameter ≥ 1/8 inch
3-A originated in the 1920s specifically for dairy processing. Today, facilities processing milk, cheese, ice cream, and many beverage products require both FDA material compliance and 3-A design certification. Artesian Systems' BUNA-N, PTFE, and Viton/FKM tri-clamp screen gasket lines carry 3-A approved materials, satisfying both requirements in a single specified component.
NSF/ANSI/CAN 61: Potable Water Contact
NSF 61 certifies materials for health effects in drinking water systems. If your beverage facility draws municipal water or your process equipment contacts a potable water supply, gaskets may need both FDA and NSF 61 certification.
NSF 61 measures contaminant leaching under simulated service conditions. It applies across:
- Water treatment plants
- Beverage production facilities using municipal supply
- Any system where gaskets contact potable water before it becomes a food ingredient
USDA Compliance: Documentation Over Evaluation
USDA FSIS does not maintain its own approved materials list. Instead, it requires manufacturers to supply a letter of guaranty confirming compliance with FDA food additive regulations. The letter must include:
- Statement of FDA compliance
- Brand/code identification
- Conditions and intended use (referencing 21 CFR 176.170 Table I food types)
- Manufacturer signature
For meat and poultry facilities under USDA inspection, procurement teams should request FDA compliance letters from gasket suppliers. Artesian Systems supplies USDA compliant gasket materials and provides FDA compliance letters upon request for federally inspected plant procurement records.
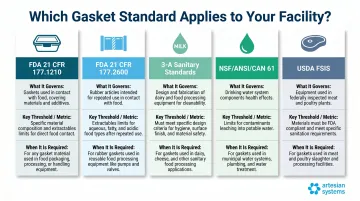
| Standard | What It Governs | Key Threshold | When Required |
|---|---|---|---|
| FDA 21 CFR 177.1210 | Container closure gaskets | 50 ppm water/alcohol extractives | Food packaging |
| FDA 21 CFR 177.2600 | Processing equipment gaskets | 20 mg/sq in water extractives | All food processing |
| 3-A Sanitary Standards | Hygienic equipment design | Ra ≤ 32 μin; CIP offset ≤ 1/32" | Dairy, beverage, food processing |
| NSF/ANSI/CAN 61 | Potable water components | Health effects criteria | Water treatment, beverage using municipal supply |
| USDA FSIS | Meat/poultry plant equipment | FDA letter of guaranty | Federally inspected meat/poultry facilities |
Food Grade Gasket Materials: A Technical Comparison
No single gasket material is universally optimal. Selection depends on operating temperature, chemical exposure (including CIP agents), pressure cycling, and food type. The following comparison establishes performance boundaries for five primary materials.
FDA Silicone Rubber
Temperature range: -67°F to 450°F (-55°C to 232°C)
Strengths:
- Widest temperature range among true elastomers
- Excellent flexibility maintains seal integrity through thermal cycling
- FDA compliant with low chemical reactivity
- Suitable for steam-in-place (SIP) processes
Limitations:
- Poor resistance to aromatic oils, strong acids, and strong alkalis
- Not recommended for CIP systems using caustic soda (NaOH) or aggressive concentrations of peracetic acid
- Unsuitable for dynamic sealing applications
Best applications: Pasteurization systems, low-acid food processing, applications requiring extreme temperature swings without aggressive chemical exposure.
EPDM (Ethylene Propylene Diene Monomer)
Temperature range: -40°F to 225°F (-40°C to 107°C)
Strengths:
- Strong resistance to water, steam, and aqueous cleaning chemicals
- Cost-effective compared to silicone and fluoropolymers
- Performs well in CIP systems with typical detergent concentrations
- Common in dairy and beverage piping
Limitations:
- Poor resistance to oils, fats, and petroleum products
- Lower maximum temperature than silicone—limits SIP capability
- Not suitable for high-fat food processing streams
Best applications: Beverage carbonation lines, dairy processing (milk, yogurt, cheese production), water-based food products, moderate-temperature CIP systems.
Artesian Systems manufactures PTFE x EPDM unitized gaskets (1.5" to 12") combining EPDM's sealing memory with PTFE's chemical resistance. This hybrid design suits systems requiring both CIP compatibility and broader chemical tolerance.
FDA Nitrile (Buna-N / NBR)
Temperature range: -40°F to 250°F (-40°C to 121°C)
Strengths:
- Excellent resistance to oils, fats, and petroleum derivatives
- Good performance with alcohols, ethers, and glycols
- Suitable for dairy suction/discharge lines and fat-carrying systems
- Cost-effective for oil-contact applications
Limitations:
- Poor steam resistance—not suitable for SIP
- Limited chemical range compared to PTFE or FFKM
- Lower temperature ceiling than silicone
Best applications: Oil processing, fat-based food products, hydraulic systems in food plants, applications involving petroleum derivatives or animal fats.
Artesian Systems offers BUNA-N screen gaskets with 3-A approval, USDA compliance, and FDA 21 CFR 177.2600/177.1550 certification in 20-micron and 100-micron configurations (1.5" to 6").
PTFE (Polytetrafluoroethylene)
Temperature range: -360°F to 550°F (-218°C to 288°C) for mineral-filled compounds
PTFE's -360°F floor is the lowest of any material in this comparison — a relevant spec for cryogenic or freeze-cycle processes.
Strengths:
- Broadest chemical resistance — near-universal compatibility
- Low friction coefficient
- Suitable for aggressive acids, solvents, and high-purity pharmaceutical processes
- FDA 21 CFR 177.1550 compliant
- Parker Hannifin Compound 0127 carries both FDA and 3-A Sanitary compliance
Limitations:
- Non-elastomeric: lacks recovery and sealing memory
- Subject to cold flow (creep) under sustained compression. This requires different joint design and potential bolt retorquing.
- Not recommended for bolted clamp assemblies
- Pressure ratings don't apply to connections 1" and above due to cold flow risk
Best applications: Aggressive chemical exposure, high-purity pharmaceutical manufacturing, wide temperature cycling, processes requiring universal chemical compatibility.
Artesian Systems supplies PTFE screen gaskets (20-micron and 100-micron) in 1.5" to 6" sizes, with 3-A approval and FDA compliance. Their PTFE x EPDM unitized gaskets reduce initial creep and cold flow compared to pure PTFE while maintaining chemical resistance.
FFKM/Perfluoroelastomer (Kalrez®)
Temperature range: -4°F to 527°F (-20°C to 275°C) continuous for Kalrez 6375
Strengths:
- Resists over 1,800 chemicals — PTFE-like inertness in elastomeric form
- True elastomer with compression set recovery
- Ideal for pharmaceutical, high-value food, and chemical processing where contamination risk is highest
- Suitable for aggressive CIP agents and extreme process conditions
Limitations:
- Cost premium: 8-35x baseline elastomer pricing (exceeds FKM fluoroelastomer cost)
- Limited low-temperature capability compared to PTFE
- Overkill for moderate conditions — reserve for applications where performance justifies cost
Best applications: Reserve FFKM for processes where a single contamination event or unplanned shutdown costs more than the material premium — pharmaceutical manufacturing with aggressive solvents, high-purity nutraceutical lines, and chemical plants cycling through extreme conditions.
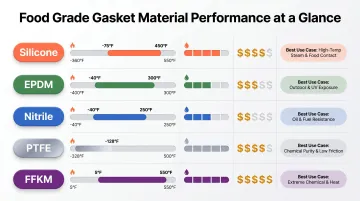
The table below summarizes key performance boundaries for quick cross-material comparison:
| Material | Temp Range | Chemical Resistance | Cost Ratio | Best Use Case |
|---|---|---|---|---|
| Silicone | -67°F to 450°F | Moderate | 2-6x | Wide temp, non-aggressive chemistry |
| EPDM | -40°F to 225°F | Water/steam/CIP | 1x (baseline) | Dairy, beverage, cost-sensitive |
| Nitrile | -40°F to 250°F | Oils/fats | 1-2x | Fat-based foods, petroleum contact |
| PTFE | -360°F to 550°F | Near-universal | 3-8x | Aggressive acids, solvents, pharma |
| FFKM | -4°F to 527°F | Universal + elastomeric | 8-35x+ | Extreme conditions, critical processes |
How to Select the Right Food Grade Gasket for Your Application
Filter 1: Chemical Compatibility
Identify every substance the gasket will contact:
- Product media (fatty foods, acidic beverages, alcoholic spirits)
- CIP cleaning agents (caustic soda, peracetic acid, chlorine compounds, phosphoric acid)
- Sanitizers (quaternary ammonium, iodophors)
- Steam (SIP cycles)
Eliminate incompatible materials:
- Silicone fails against aromatic oils and strong caustic
- EPDM fails against oils, fats, and petroleum products
- Nitrile fails against steam and limited chemical range
- PTFE handles nearly everything but lacks elastomeric recovery
- FFKM tolerates all of the above but at premium cost
Example decision: A dairy plant processing whole milk (high fat content) with daily caustic CIP cycles requires a material resistant to both fats and caustic. EPDM fails on fat resistance; silicone fails on caustic; nitrile fails on steam/CIP. Solution: PTFE or PTFE x EPDM unitized gasket.
Filter 2: Operating Temperature and Pressure
Determine actual operating range, not just nominal:
- Process temperature during production
- CIP temperature (typically 140-180°F)
- SIP temperature (250-285°F steam)
- Ambient low temperature (cold storage, outdoor piping)
Apply safety factor: Don't operate at material's maximum rated temperature. Target 80% of maximum for continuous service to prevent premature compression set.
Example: If your process includes 270°F SIP cycles, EPDM (max 225°F) is eliminated. Silicone (450°F max) operates at 60% of rated capacity—acceptable. PTFE (550°F max) operates at 49% of rated capacity—excellent margin.
Pressure cycling: Gaskets experiencing frequent pressure changes develop compression set faster. High-cycle applications favor materials with better recovery (EPDM, FFKM) or require more frequent replacement schedules for PTFE.
Filter 3: Regulatory Requirement Matching
Map your facility type to required certifications:
| Facility Type | Required Standards |
|---|---|
| Dairy processing | FDA 21 CFR 177.2600 + 3-A Sanitary |
| Beverage (municipal water) | FDA 21 CFR 177.2600 + NSF/ANSI 61 |
| Meat/poultry (USDA inspected) | FDA 21 CFR 177.2600 + USDA letter of guaranty |
| Pharmaceutical | FDA 21 CFR 177.2600 + USP Class VI (if applicable) + ASME BPE (recommended) |
| General food processing | FDA 21 CFR 177.2600 minimum |

Critical: FDA compliance alone doesn't satisfy 3-A requirements. 3-A governs surface finish (Ra ≤ 32 μin) and joint geometry (offset ≤ 1/32") that FDA doesn't address. Dairy and many beverage facilities require both.
Filter 4: Dimensional and System Design
Gasket type selection:
- Standard tri-clamp ring: Most common for sanitary systems; simple replacement
- Envelope/unitized (PTFE x EPDM): Reduces creep vs. pure PTFE; accommodates slight ferrule misalignment; better for frequent replacement under GMP
- Screen gaskets: Inline filtration (5, 20, 100 micron) + sealing in one component
Tri-clamp sizing: Gasket size is determined by tube OD, not flange OD. Standard sizes: 1.5", 2", 2.5", 3", 4", 6", 8", 10", 12". Follow ISO 2852, DIN 32676, or BS 4825-3 dimensional standards.
Surface finish requirements: 3-A requires Ra ≤ 32 μin. If your existing fittings don't meet this, upgrading gasket material alone won't achieve 3-A compliance—fitting replacement or electropolishing may be required.
For multi-material systems, custom geometries, or applications requiring both extreme chemical resistance and specific dimensional tolerances, working with a sanitary component specialist reduces specification errors before they reach production. Artesian Systems provides custom engineering services — including CNC machining, process system design, and material selection consultation — for pharmaceutical, nutraceutical, and food processing applications.
Common Food Processing Applications by Industry
Dairy and Beverage Processing
3-A Sanitary Standards originated in the 1920s specifically for milk pipe fittings — and for good reason. Dairy operations carry the highest bacterial contamination risk of any food category, because protein and fat content create ideal growth conditions inside improperly cleaned joints.
Material selection for dairy and beverage lines:
- EPDM: Most common for milk lines, pasteurization heat exchangers, and water-based beverage filling due to cost-effectiveness and CIP compatibility
- Silicone: Preferred for high-temperature pasteurization (HTST, UHT systems) where the wider temperature range is needed
- PTFE/EPDM unitized: Growing adoption where both CIP compatibility and fat resistance are required simultaneously
Gaskets in these systems must survive daily thermal cycling — from ambient to 180°F during CIP, up to 270°F during SIP — plus repeated exposure to alkaline detergents and acid rinses. EPDM handles moderate CIP well. Silicone is required for SIP cycles. PTFE is the call when chemical concentrations get aggressive.

Artesian Systems supplies 3-A approved screen gaskets and unitized gaskets sized for standard dairy and beverage tri-clamp connections (1.5" to 12"), with materials meeting FDA, 3-A, and USDA requirements.
Pharmaceutical and Nutraceutical Manufacturing
Pharmaceutical processes are among the most demanding sealing environments: aggressive solvents (methanol, ethanol, acetone, hexane), strict purity requirements around extractables, and wide temperature swings. PTFE handles this through near-universal chemical resistance; FFKM adds elastomeric recovery for dynamic seals where PTFE's rigidity creates a limitation.
Certification tiers that matter in pharma:
- FDA 21 CFR compliance: Establishes base food-contact safety for materials
- USP Class VI: Measures biological reactivity through in-vivo tests (systemic injection, intracutaneous reactivity, implantation) at 50°C, 70°C, and 121°C — both certifications are often required simultaneously
- ASME BPE-2022: Sets hygienic design requirements including gasket location between coupling faces and process-side intrusion limits
Artesian Systems' Viton/FKM and PTFE screen gaskets have passed USP Class VI Cytotoxicity testing — relevant for facilities where both FDA 21 CFR and USP Class VI are specified on the same drawing.
Most state cannabis programs require food-grade equipment, and extraction systems running heptane, hydrocarbons, or CO2 demand broad chemical resistance across the full solvent range. PTFE-based gaskets are the standard specification here. Artesian Systems' founder developed a patented medical hydrocarbon extractor, and the company supplies gaskets rated for extraction equipment handling these solvents.
Meat, Poultry, and Dry Food Processing
FSIS Directive 7310.5 names gaskets and O-rings directly as rubber contamination sources. Foreign material between 7mm and 25mm is considered high choking/injury risk — triggering immediate enforcement actions including noncompliance records, product retention, recalls, and potential operation suspension. That regulatory specificity shapes every material decision in this sector.
Material selection:
- Nitrile (BUNA-N): Preferred for meat processing due to excellent fat and oil resistance at a practical cost
- Specially formulated EPDM: Used in some meat operations where enhanced oil resistance compounds are available
Metal-detectable gaskets incorporate 316L stainless steel directly into the gasket compound, making fragments visible to standard metal detectors and X-ray systems. This matters most for dense products like peanut butter, where conventional metal detection loses sensitivity. The peanut butter manufacturer referenced in the introduction ended their recall exposure by switching to Detectomer Tuf-Steel gaskets — a PTFE/316L stainless blend.
Artesian Systems offers USDA-compliant BUNA-N and Viton/FKM gaskets for meat and poultry processing. For facilities evaluating detectable gasket technology, their engineering team can identify the right compound for the specific product and detection system in use.
Installation, Maintenance, and When to Replace Food Grade Gaskets
Proper Installation Practices
Pre-installation:
- Inspect gasket for damage, contamination, or dimensional defects before installation
- Clean gasket contact surfaces to Ra ≤ 32 μin (for 3-A compliance) and remove all debris, old gasket material, and product residue
- Never reuse gaskets removed during maintenance in sanitary systems—single-use installation only
Torque sequences for flanged joints:
- Use star pattern (cross-pattern) tightening sequence
- Apply torque in 3-4 stages, incrementally increasing to final specification
- Avoid over-tightening PTFE gaskets—excessive compression accelerates cold flow
- For PTFE gaskets in critical applications, plan for retorquing after initial compression set (typically 24-48 hours after installation)
Tri-clamp installations:
- Ensure ferrules are properly aligned with offset ≤ 1/32" (3-A requirement)
- Check gasket seats flush without bulging or extrusion
- For unitized/envelope gaskets, verify EPDM core faces process side for proper sealing memory
- Avoid stretching gaskets beyond 50% of diameter during installation to prevent pockmarking damage
Scheduled Inspection and Maintenance
Visual inspection at each maintenance cycle:
- Compression set: Permanent deformation preventing gasket recovery—indicates replacement needed
- Surface cracking: Fine cracks indicate material degradation from chemical attack or thermal cycling
- Discoloration: May indicate chemical incompatibility or beginning of degradation
- Swelling: Indicates solvent absorption—wrong material for application
- Loss of seal: Product weeping, steam leaks, or pressure loss
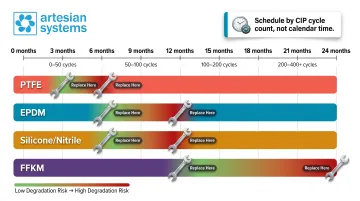
Align inspection intervals with CIP cycles rather than calendar dates. High-frequency CIP operations — especially those using aggressive chemistry like high-caustic concentrations or peracetic acid — accelerate degradation faster than time alone predicts.
For valve seat gaskets and diaphragms, follow OEM service intervals without exception. Extending diaphragm life beyond rated operating hours risks premature failure and contamination.
When to Replace Immediately vs. Scheduled Replacement
Immediate replacement triggers:
- Any contamination event where gasket is identified as potential source
- Visible damage (cracking, tearing, extrusion, pockmarking)
- Failed pressure test or leak detection
- Process chemistry or temperature change exceeding gasket's original specification
- Swelling or significant compression set observed during inspection
- After any maintenance event that required gasket removal
Scheduled replacement strategy:
- Establish preventive replacement schedules based on CIP cycle counts, not calendar time alone
- Track cycles by material type: PTFE may require replacement every 3-6 months under high-CIP frequency; EPDM may last 6-12 months; FFKM may extend 12-24 months
- Document gasket replacement in FSMA and HACCP preventive controls programs
- Maintain gasket replacement logs as part of sanitary equipment maintenance records
At critical process points, scheduled replacement is nearly always the lower-cost path. A $20 gasket replaced on a preventive schedule costs a fraction of what a single contamination event triggers in downtime, product loss, and potential recall exposure.
Frequently Asked Questions
What is the difference between a food grade gasket and a standard industrial gasket?
Food grade gaskets are manufactured from materials specifically approved for food contact: non-toxic, odorless, tasteless, and non-contaminating per FDA 21 CFR testing. Standard industrial gaskets may contain fillers, plasticizers, or compounding agents that are prohibited in food applications because they can leach harmful substances into the product stream.
What does FDA 21 CFR 177.1210 mean for gasket compliance?
This regulation governs closure-sealing gaskets for food containers, setting maximum extractive limits (50 ppm water/alcohol, 50-500 ppm heptane depending on type) to ensure no harmful substances migrate into food. It applies to jar lids, bottle caps, and container seals. Processing equipment gaskets fall under a separate regulation: 21 CFR 177.2600.
Are silicone gaskets better than EPDM for food processing applications?
Neither is universally superior. Silicone handles wider temperature ranges (-67°F to 450°F) and is required for SIP applications, while EPDM performs better with aqueous CIP chemicals at moderate temperatures (up to 225°F) and costs less. The right choice depends on your specific process temperature profile and chemical exposure.
Do food grade gaskets need to meet 3-A standards as well as FDA approval?
FDA approval covers material safety (what the gasket is made of). 3-A Sanitary Standards govern design and cleanability (surface finish Ra ≤ 32 μin, joint geometry with offset ≤ 1/32"). Dairy facilities and many beverage operations must meet both standards; FDA material compliance alone is insufficient for 3-A certification.
How often should food grade gaskets be replaced?
Inspect gaskets at every maintenance cycle and replace at any sign of compression set, cracking, discoloration, or swelling. Replacement frequency varies by material, temperature cycling, and CIP/SIP exposure — scheduling replacements by CIP cycle count rather than calendar time reduces contamination risk more reliably.
Can the same gasket material be used for both food and pharmaceutical applications?
PTFE and FFKM are commonly used across both sectors due to broad chemical resistance and low extractables. Pharmaceutical applications typically require additional documentation: FDA Drug Master File (DMF), USP Class VI testing, and ASME BPE compliance. Verify all certifications with your gasket supplier before finalizing the specification.
Need help selecting the right food grade gasket for your specific process? Artesian Systems manufactures FDA 21 CFR 177.2600 and 177.1550 compliant tri-clamp gaskets (1.5" to 12") in EPDM, PTFE, and Viton/FKM materials, with custom engineering services for complex applications. Contact the technical team at +1 801-318-4079 or artesiansystems@gmail.com for specification support.


