: What's Available?](https://file-host.link/website/artesian-systems-s8v6l9/assets/blog-images/6a327570-beef-44a4-9dc8-5a7b01c9e83d/1778486264889546_a4702c4000864f6ea2a4573d53e6d6cb/360.webp)
Introduction
In pharmaceutical, biopharmaceutical, food processing, and cannabis extraction environments, not all sanitary gaskets are created equal. Using the wrong material can trigger compliance failures, introduce contamination risk, or cause costly downtime.
USP Class VI certification is the dividing line between gaskets acceptable for general sanitary use and those qualified for high-purity, biocompatible applications. This distinction matters because gaskets form the sealing point between process fluids and system components. Any leachable compound from a non-compliant gasket becomes a direct contamination pathway.
What follows covers what USP Class VI means for sanitary gaskets, which material types are commercially available, and how to match the right material to your process requirements.
TL;DR
- USP Class VI requires materials to pass three in vivo biological reactivity tests—a stricter standard than FDA food-grade approval
- Buna-N (nitrile) and white peroxide-cured silicone are FDA-approved only and do NOT meet USP Class VI standards
- Compliant materials include platinum-cured silicone, PTFE, EPDM, FKM, envelope gaskets, and FFKM (Kalrez)
- Material selection depends on process fluid chemistry, cleaning method, temperature range, and regulatory documentation requirements
- FFKM offers the broadest chemical resistance but at a significant cost premium over PTFE or silicone options
What Is USP Class VI and Why Does It Matter for Sanitary Gaskets?
USP Class VI represents the most stringent plastics and elastomers classification under USP General Chapter <88>, titled "Biological Reactivity Tests, In Vivo." The United States Pharmacopeia (USP)—a non-government standards organization—publishes this chapter as part of its 2024 edition.
The Three Required Tests
To achieve Class VI certification, materials must pass all three in vivo animal tests:
- Systemic Injection Test – Measures systemic toxicity in mice via IV or IP injection of material extracts; passing requires no deaths, no abnormal behavior in two or more mice, and body weight loss not exceeding 2g in three or more mice
- Intracutaneous Test – Evaluates local tissue irritation in rabbits through intradermal injection; the difference between sample and control scores for erythema/edema must be 1.0 or less
- Implantation Test – Assesses tissue reaction in rabbits through surgical placement in paravertebral muscle; the difference between sample and control encapsulation scores must not exceed 1.0

Class VI is the only classification requiring both the full four-media extraction panel (NaCl, alcohol/NaCl, PEG 400, vegetable oil) and the implantation test. Manufacturers prepare material extracts under one of three conditions: 50°C for 72 hours, 70°C for 24 hours, or 121°C for 1 hour — the autoclave condition simulating steam sterilization exposure.
Why This Matters for Gaskets
Sanitary gaskets form the sealing interface between fluid-contact surfaces in Tri-Clamp, RJT, and similar fittings. In pharmaceutical, bioprocessing, and high-purity food applications, any extractable compound that leaches from the gasket into the process stream represents a contamination risk and a regulatory liability.
USP Class VI testing validates that leaching risk falls within acceptable limits through biological reactivity assessment, not just chemical composition analysis. That distinction matters: the standard is not a regulatory mandate on its own, but it is industry-expected in biopharmaceutical, nutraceutical, and certain food and beverage contexts.
USP Class VI certification adds to FDA CFR 21 requirements rather than replacing them. Artesian Systems sources sanitary components compliant with the following standards, providing a documented compliance baseline for high-purity process systems:
- FDA 21CFR177.2600 and 1550 — food-contact elastomers and plastics
- 3A Sanitary Standards — hygienic equipment design for dairy and food processing
- USDA — materials approved for use in federally inspected facilities
FDA Approval vs. USP Class VI: Understanding the Difference
FDA 21 CFR 177.2600 governs rubber articles intended for repeated food contact. It operates as an ingredient allowlist: rubber must be prepared from specifically listed polymers and adjuvant substances (vulcanization agents, antioxidants, plasticizers, and similar additives).
The regulation sets extraction limits — not exceeding 20 mg/sq inch in distilled water and 175 mg/sq inch in n-hexane — but includes no in vivo biocompatibility testing requirements.
Similarly, FDA 21 CFR 177.1550 covers perfluorocarbon resins like PTFE for food-contact applications. It specifies identity/purity requirements and extraction limits (max 0.2 mg/sq inch total extractives; max 0.03 mg/sq inch fluoride extractives) but contains no biological testing component.
The Key Distinction
FDA compliance addresses material safety through chemical composition and extraction chemistry. USP Class VI addresses biocompatibility through animal-model biological reactivity testing. As industry experts confirm, biopharmaceutical gaskets must meet FDA standards and pass USP Class VI testing. Neither standard substitutes for the other.
FDA-Only Materials Not Suitable for USP Class VI
Two common sanitary gasket materials carry FDA approval but fail USP Class VI testing:
- Buna-N (Nitrile) – The least expensive sanitary gasket material, widely stocked and FDA-compliant, but not USP Class VI certified
- White Peroxide-Cured Silicone – FDA-approved and commonly used, but lacks USP Class VI certification; often confused with platinum-cured silicone, which does pass Class VI
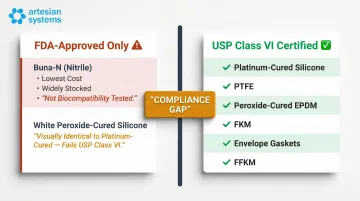
If your facility requires USP Class VI documentation for incoming components, FDA-only materials will fail qualification. Catching this gap at the design or procurement stage prevents costly requalification later.
USP Class VI Sanitary Gasket Materials: What's Available
Five material categories represent the sanitary gasket options commercially available with USP Class VI certification. Size availability and specific grades vary by manufacturer—always request certification documentation to verify compliance for your specific application.
Platinum-Cured Silicone
What it is: Silicone gaskets cured using a platinum catalyst rather than peroxide. The platinum cure process uses addition polymerization, leaving little to no residual byproducts in the cured material.
Peroxide curing, by contrast, decomposes organic peroxides under heat—leaving behind residual byproducts that cause odors, discoloration, and outgassing. That's why platinum-cured silicone passes USP Class VI while standard white (peroxide-cured) silicone typically does not.
Best-suited applications:
- Biopharmaceutical process piping
- Vaccine and parenteral drug manufacturing
- Nutraceutical processing
- Applications requiring repeated steam sterilization (SIP) or autoclave cycles
- Commercially available in Tri-Clamp sizes ½" through 12"
Temperature rating: -40°F to 450°F (-40°C to 232°C) for continuous service with SIP exposure rated for cycles up to 30 minutes.
Key trade-offs:
- Limited resistance to oils, fats, and hydrocarbon-based solvents
- Not appropriate for cannabis hydrocarbon extraction or fatty media processing
- Higher cost than EPDM or Buna-N
- Must specifically verify platinum-cure formulation—white silicone gaskets look identical but may be peroxide-cured and FDA-only
PTFE (Teflon)
What it is: Polytetrafluoroethylene (PTFE) gaskets are chemically inert, non-elastomeric seals that provide rigid sealing between tri-clamp ferrule faces. Most PTFE sanitary gaskets carry USP Class VI certification alongside FDA 21 CFR 177.1550 and 3-A Sanitary Standard 18-03 compliance.
Best-suited applications:
- Aggressive solvent environments
- High-temperature processes
- Pharmaceutical and chemical manufacturing where broad chemical resistance is a hard requirement
- Commercially available in sizes ½" through 6"
Temperature rating: -100°F to 500°F (-38°C to 260°C)—the widest usable range among common sanitary gasket materials.
Key trade-offs:
- PTFE has low resilience and does not compress like elastomers—proper ferrule alignment and torque are critical
- Prone to cold flow/creep over time under sustained compression
- Even at room temperature, PTFE experiences cold flow, which can cause progressive seal loosening
- Not recommended for systems with large temperature variations or vibration
- May require more frequent inspection in thermally cycled systems
- Requires high-pressure clamps for proper sealing in SIP applications
Peroxide-Cured EPDM
What it is: Ethylene propylene diene monomer (EPDM) gaskets using a peroxide cure system. Unlike sulfur-cured EPDM (which is FDA-approved only), peroxide-cured EPDM meets USP Class VI criteria.
Sulfur vulcanization leaves behind residual sulfur-containing extractables shown to alter process fluid integrity and negatively affect mammalian cell yields. Peroxide curing avoids those byproducts entirely.
Best-suited applications:
- Biopharmaceutical systems requiring intermittent steam sterilization (SIP)
- Water-for-injection (WFI) systems
- Clean steam service
- Commercially available in sizes ½" through 12"
Temperature rating: -30°F to 300°F (-34°C to 149°C); some manufacturers rate up to 310°F (154°C) with dimensional stability for repetitive SIP cycles.
Key trade-offs:
- Does not perform well with fats, oils, or hydrocarbon-based solvents
- Not suitable for dairy fat service or extraction applications
- Rated "Not Recommended" for hydrogen peroxide CIP service
- Engineers must verify peroxide-cured (not sulfur-cured) variant when USP Class VI documentation is required
Viton / FKM (Fluoroelastomer)
What it is: Fluorocarbon elastomer (FKM) gaskets—commonly sold under the Viton brand name—combine the elasticity and sealing performance of traditional rubber with high chemical resistance. Most FKM sanitary gaskets commercially available today carry USP Class VI certification, but this is compound-specific.
Best-suited applications:
- High-temperature service
- Aggressive chemical environments including acids, solvents, and fuels
- Cannabis and hemp hydrocarbon extraction systems
- Applications requiring both chemical resistance and reliable gasket compression
- Commercially available in Tri-Clamp sizes ½" through 12"
Temperature rating: -30°F to 400°F (-34°C to 204°C) for standard grades; specialty grades like Viton Extreme (ETP) handle up to 450°F (232°C) continuous and 600°F (315°C) short-term.
Key trade-offs:

- FKM costs more than silicone or EPDM
- Standard bisphenol-cured FKM degrades rapidly in continuous steam; rated unsuitable for continuous SIP
- Standard FKM has poor resistance to bases/alkalies
- Viton Extreme (ETP) grades offer excellent steam and CIP resistance (including NaOH, HNO3, H3PO4) but at higher cost
- Not all FKM grades or colors carry USP Class VI—always request certificate of conformance specific to the lot
- USP Class VI certification applies to the specific compound formulation, not to FKM as a material class
Specialty Options: Envelope Gaskets, PTFE/SS Blends, and Perfluoroelastomers
Envelope gaskets: An outer PTFE shell encapsulating an inner core of EPDM or FKM. The combination provides chemical inertness and non-stick surface of PTFE on the process side while the elastomeric core delivers compression resilience that solid PTFE lacks. Available in sizes ½" through 12" and generally USP Class VI compliant. Recommended as alternatives to solid PTFE where fitting misalignment exists.
PTFE/316 SS blends (Tuf-Steel): Gaskets molded from PTFE resin sintered with stainless steel particles. These deliver excellent chemical and thermal resistance with USP Class VI compliance. Rated at -320°F to 550°F (-195°C to 288°C)—the widest range of any sanitary gasket material. That range, combined with high purity and long service life, makes them well-suited for thermally demanding processes and aggressive chemical service.
Perfluoroelastomers (FFKM/Kalrez): Materials like DuPont Kalrez offer near-PTFE chemical compatibility with the sealing elasticity of a rubber gasket. All Kalrez Life Sciences grades carry USP <87> and <88> Class VI certification at 121°C, along with FDA compliance and 3-A certification. Maximum temperatures range from 220°C to 260°C (428°F to 500°F) depending on grade. These are the highest-performing (and most expensive) option, appropriate for demanding solvent or pharmaceutical processes where aggressive solvents or elevated temperatures rule out silicone, EPDM, and standard FKM.
How to Choose the Right USP Class VI Gasket for Your Application
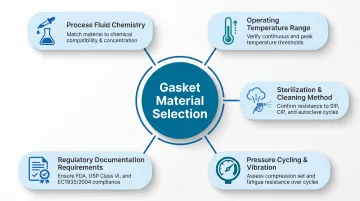
Material selection must be driven by process requirements—not by default preferences or lowest cost. Five key variables determine the right gasket material:
Critical decision factors:
- Process fluid chemistry – Aqueous vs. solvent vs. fatty/oil-based
- Operating temperature range – Continuous and peak exposure temperatures
- Sterilization or cleaning method – CIP chemistry, SIP steam temperature, autoclave cycles
- Pressure cycling and vibration – Systems with thermal or mechanical stress
- Regulatory documentation requirements – USP Class VI, FDA, 3A, USDA compliance levels
Decision-Tree Approach
For steam sterilization + aqueous pharma media: Start with peroxide-cured EPDM or platinum-cured silicone. Both handle repetitive SIP cycles and provide biocompatibility in WFI and clean steam service.
For aggressive solvents or hydrocarbon extraction (cannabis/hemp): FKM or FFKM are appropriate. Standard silicone and EPDM degrade in oils and hydrocarbon environments. Viton/FKM provides good chemical resistance at moderate cost; FFKM (Kalrez) handles the broadest solvent resistance, including ketones, esters, and aromatic hydrocarbons.
When chemical resistance is the top priority: PTFE or envelope gaskets deliver broad chemical compatibility across most acids, bases, and solvents. Accept the trade-off: PTFE lacks resilience and is prone to cold flow, so proper installation and inspection protocols are critical.
The Curing Method Check for Silicone
When specifying silicone gaskets, buyers must confirm platinum-cure, since not all white silicone is USP Class VI compliant. White peroxide-cured silicone and platinum-cured silicone look identical but only platinum-cured passes Class VI testing. Always verify the cure method in procurement specifications.
Documentation Requirements
USP Class VI certification is a per-compound, per-lot certification. Require a certificate of conformance (CoC) and, where available, the actual test report. Confirm that any gasket material also aligns with other applicable standards for your industry—3A, USDA, or FDA CFR 21.
Artesian Systems sources sanitary components that comply with 3A, USDA, and FDA 21CFR177.2600 and 1550 standards, providing a documented compliance baseline for high-purity process systems alongside USP Class VI qualification where required.
CIP Chemistry Compatibility
Compliance with product contact requirements doesn't guarantee CIP compatibility. A material that handles your process fluid can still degrade under aggressive cleaning agents — check both separately:
| CIP Chemical | PTFE | Viton Extreme (ETP) | Silicone | EPDM |
|---|---|---|---|---|
| NaOH (Caustic) | Excellent | Excellent | Limited | Excellent |
| Nitric Acid | Excellent | Excellent | Limited | — |
| Phosphoric Acid | Excellent | Excellent | — | — |
| Hydrogen Peroxide | Excellent | — | — | Not Recommended |
Partial data shown; consult manufacturer for complete charts. Source: Process Technologies Viton Extreme Data Sheet
EPDM is often preferred for steam/water-based CIP while FKM may be necessary where solvent-based or oxidizing cleaning is used.
What to Check Before Finalizing Your Gasket Selection
Never Select Based on Price Alone
Buna-N is the least expensive sanitary gasket material and widely stocked, but it is not USP Class VI certified. Using it in a validated pharmaceutical or bioprocessing system creates a compliance gap that can trigger requalification or batch rejection.
Verify Grade Specificity
- EPDM: Available in sulfur-cured (FDA-only) and peroxide-cured (USP Class VI) versions
- FKM: Grades vary in chemical resistance depending on fluorine content and cure system
- Silicone: Platinum-cure vs. peroxide-cure is the dividing line for USP Class VI compliance

Always request the specific compound designation and certificate, not just the material name.
Take a System-Level View
Gasket material selection should account for the full process lifecycle:
- Installation frequency and ease of replacement
- Chemical exposure during CIP/SIP cycles
- Thermal fatigue from steam cycles
- Cost of unplanned changeouts due to material failure
A higher upfront cost for the right material saves on downtime and compliance remediation. Over multiple production cycles, that difference compounds — especially in validated systems where a single nonconformance can halt an entire batch.
Conclusion
USP Class VI sanitary gaskets span a meaningful range of materials, each with different chemical compatibility profiles, sterilization tolerances, and application fits:
- Platinum-cured silicone — broad temperature range, steam-sterilizable, common across pharma and food
- PTFE — chemically inert, ideal for aggressive solvents and oxidizers
- Peroxide-cured EPDM — strong CIP/SIP performance, cost-effective for aqueous systems
- FKM (Viton) — superior chemical resistance for oils, fuels, and hydrocarbon processes
- Envelope and FFKM gaskets — specialty constructions for extreme chemical or temperature demands
The certification confirms low-extractables biocompatibility, but the right material choice depends on your process.
The distinction between FDA-compliant and USP Class VI certified materials is consequential — each addresses different regulatory requirements, and confusing them creates real compliance risk. Suppliers should be able to provide documented material compliance across USP Class VI, FDA 21CFR, 3A, and USDA standards. Artesian Systems manufactures tri-clamp gaskets meeting FDA 21CFR177.2600/1550, 3A, and USDA requirements, and can help match the right certified material to your process demands.
Frequently Asked Questions
What does USP Class VI mean?
USP Class VI is the most rigorous elastomer/plastic classification under USP General Chapter <88>, requiring materials to pass three in vivo biological reactivity tests (systemic injection, intracutaneous, and implantation) to confirm that toxic extractables are absent at specified temperature conditions.
What are the USP Class VI requirements for gaskets?
Gasket materials must pass three Chapter <88> in vivo tests at multiple temperature conditions (up to 121°C autoclave exposure). Manufacturers must provide documented test results and a certificate of conformance; certification applies to the specific compound formulation, not just the generic material category.
Which gasket materials meet USP Class VI?
The main commercially available USP Class VI-certified sanitary gasket materials are: platinum-cured silicone, PTFE, peroxide-cured EPDM, FKM (Viton), envelope gaskets (PTFE-over-EPDM or FKM), PTFE/316 SS blends (Tuf-Steel), and perfluoroelastomers (FFKM/Kalrez).
Is EPDM USP Class VI?
Peroxide-cured EPDM is USP Class VI certified and widely used in biopharma steam service. Sulfur-cured EPDM is FDA-approved only and not USP Class VI compliant. The distinction lies in the cure system, not the base polymer—buyers must confirm the correct grade.
How do Viton (FKM) and Buna-N (nitrile) differ for gasket use?
FKM offers far greater chemical and thermal resistance and is available with USP Class VI certification, making it suitable for pharmaceutical and bioprocessing applications. Buna-N is the most economical option but is FDA-approved only and not suitable for USP Class VI-required applications.
What is the difference between USP Class VI and ISO 10993?
USP Class VI uses in vivo animal tests under Chapter <88> to evaluate acute biocompatibility through three fixed tests. ISO 10993 is a broader international framework for medical device qualification, covering additional endpoints—such as genotoxicity and chronic toxicity—using a risk-based test selection matrix rather than a fixed battery.


