
Introduction
In food processing, a non-compliant or failing seal isn't just a maintenance issue—it's a contamination risk, a regulatory violation, and a brand liability. The stakes are measured in millions of pounds: in 2022, USDA FSIS foreign-material recalls—including rubber gasket fragments—were triple those tied to toxic E. coli. Tyson alone recalled nearly 12 million pounds of chicken in 2019 due to foreign material contamination. The seal at the food-contact surface is often the first line of defense against these failures.
PTFE seals have become the industry standard because they address this challenge at the material level. Chemically inert, thermally stable, and resistant to aggressive cleaning cycles, PTFE handles what most elastomers can't.
This guide covers the FDA regulatory framework, key material properties, seal types, CIP/SIP compatibility, and a practical selection framework to help you specify the right seal for your operation.
TLDR:
- PTFE's chemical inertness prevents food contamination and survives aggressive CIP/SIP cycles (caustic cleaners, acids, steam up to 260°C)
- FDA 21 CFR 177.1550 covers virgin PTFE; filled grades require independent filler verification under FDA regulations
- 3A certification (launched January 2026 for seal replacement parts) addresses hygienic design beyond material compliance
- Spring-energized and envelope gasket designs compensate for PTFE's cold-flow limitation under sustained load
- Proper seal selection means matching seal type, material grade, and regulatory credentials to your specific application and sanitation protocol
Why PTFE Is the Preferred Material for Food-Grade Seals
Chemical Inertness: The Defining Food-Safety Advantage
PTFE's molecular structure—consisting of carbon atoms fully shielded by fluorine—makes it one of the most chemically inert materials available. Unlike elastomers or plastics that can absorb flavor compounds, preservatives, or cleaning agents, PTFE neither absorbs food media nor leaches plasticizers or additives back into the product. That means no flavor carry-over between batches and no risk of seal-derived contamination reaching the final product.
Chemical resistance testing rates PTFE as fully compatible with beer, fruit juices, milk products, sugars, syrups, fats, acids, and alkalis at temperatures ranging from 20°C to 100°C. The material is affected only by molten alkali metals and rare fluorinated compounds at extreme temperatures and pressures—conditions far outside food processing parameters.
High-Temperature Resistance for Sterilization and Hot-Fill Processing
PTFE maintains seal integrity across a continuous service range of -240°C to 260°C (-400°F to 500°F), with a melting point of 327°C (621°F). This thermal stability is critical for:
- Steam sterilization (SIP): Withstands superheated steam at 121-150°C without degradation
- Hot-fill processing: Maintains dimensional stability during filling operations at elevated temperatures
- Thermal cycling: Resists repeated temperature shifts between production and sanitation cycles
According to manufacturer datasheets, virgin PTFE has been tested to 500°F for 20,000 hours per UL 746B standards, confirming long-term thermal reliability.
Broad Chemical Resistance Across Production and Sanitation Cycles
Food processing equipment must perform across two distinct chemical environments: production (food media) and sanitation (aggressive cleaning agents). PTFE handles both.
Production media:
- Fats, oils, and greases
- Organic acids (citric, acetic, lactic)
- Alcohols and ethers
- Sugar solutions and glycols
Sanitation is where many elastomers fail — PTFE handles these cleaning agents equally well:
Sanitation agents:
- 1-3% sodium hydroxide (NaOH) at 65-80°C for protein and fat removal
- 0.5-1.5% nitric acid (HNO₃) at 55-65°C for mineral scale removal
- Sodium hypochlorite (chlorine-alkaline media)
- Peracetic acid and hydrogen peroxide disinfectant mixtures
Most elastomeric seal materials degrade through volume change, hardness shifts, and tensile strength loss under repeated CIP exposure.
Low-Friction Surface for Clamp-Style Sanitary Fittings
PTFE's coefficient of friction is 0.1 dynamic per ASTM D1894—one of the lowest of any solid material. Combined with a Shore D hardness of 50-65 (ASTM D2240), PTFE compresses and conforms under the relatively low clamping forces typical of tri-clamp and sanitary flange connections.
This matters because sanitary piping systems rely on quick-release clamps rather than heavily bolted flanges. The seal must achieve reliable contact without requiring high torque or load, and PTFE's combination of softness and low friction makes this possible.
Cold Flow: The Known Limitation and Engineering Solutions
PTFE exhibits "cold flow" or creep—a permanent deformation under sustained compressive load, even at room temperature. Unlike rubber elastomers with cross-linked polymer chains that provide elastic recovery, PTFE's uncross-linked chains slide past each other permanently under pressure. This causes gasket thinning, loss of bolt clamping force, and eventual leaks.
Engineering mitigations include:
- Filled PTFE grades (glass, carbon, or bronze): Fillers create a rigid internal matrix that resists chain sliding — each filler must independently meet FDA compliance requirements
- Spring-energized seals: An internal spring applies continuous radial or axial force, compensating for PTFE's lack of elastic memory without relying on bolt load alone
- Envelope gaskets: A PTFE jacket over an EPDM core delivers PTFE's chemical inertness on the contact face while the rubber core provides the compressibility and recovery PTFE lacks
- Re-torquing schedules: Periodic bolt re-tightening restores clamping force lost to creep — especially important in high-temperature flanged connections
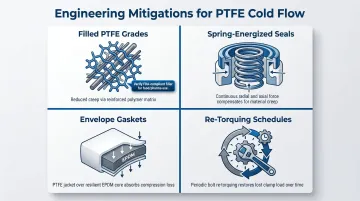
For high-temperature or high-load applications, specify one of these mitigations upfront — cold flow accelerates with both variables, and a seal failure discovered during production is far more costly than the upgrade.
Decoding FDA Compliance: 21 CFR 177.1550, 177.2600, and Beyond
21 CFR 177.1550: Virgin PTFE for Repeated Food Contact
FDA regulation 21 CFR 177.1550 specifically covers perfluorocarbon resins—including virgin PTFE homopolymer—approved for articles intended for repeated food contact. This regulation is the baseline reference point when specifying PTFE seals for food-contact surfaces.
Key specifications:
| Parameter | Limit |
|---|---|
| Total extractives | ≤ 3.1 mg/sq dm (0.2 mg/sq in) |
| Fluoride extractives | ≤ 0.46 mg/sq dm (0.03 mg/sq in) |
| Melt viscosity (Type 1 PTFE) | ≥ 10⁴ poises at 380°C |
| Thermal instability index | ≤ 50 |
Extraction testing uses distilled water, 50% ethanol, n-heptane, and ethyl acetate at reflux for 2 hours. These tests verify that PTFE does not leach harmful substances into food products under normal use conditions.
21 CFR 177.2600: Rubber Articles for Repeated Use
This regulation applies to compounded rubber and elastomeric articles—including elastomeric seal components—using a "positive list" approach. Every ingredient (base polymer, curing agents, fillers, colorants) must be explicitly listed in the regulation's ingredient table.
Extraction limits:
| Food Type | Extractant | Temp | Limit (First 7 hr) | Limit (Next 2 hr) |
|---|---|---|---|---|
| Aqueous food | Distilled water | Reflux | 20 mg/sq in | 1 mg/sq in |
| Fatty food | n-Hexane | Reflux | 175 mg/sq in | 4 mg/sq in |
This regulation is relevant to envelope PTFE gaskets (where PTFE is bonded to an elastomeric core) and to filled PTFE compounds containing elastomeric additives.
The Filler Compliance Nuance: Virgin Approval Does Not Extend Automatically
Critical principle: FDA approval of virgin PTFE under 177.1550 does not automatically extend to filled or compounded grades. Any PTFE seal that includes pigments, adhesive backings, reinforcement fibers, or fillers must have each additive independently verified as FDA-compliant.
For example, a 25% glass-filled PTFE gasket requires:
- Virgin PTFE compliance under 177.1550
- Independent verification that the glass fiber is approved for food contact
- Confirmation that any binders or sizing agents on the glass are also compliant
When specifying filled PTFE seals, always request full material compliance documentation from suppliers. Virgin PTFE's inherent compliance is an advantage that disappears the moment fillers are added.

3A Sanitary Standards: Hygienic Design Beyond Material Compliance
FDA regulations address material safety through extraction limits and compositional requirements. 3A Sanitary Standards add a second critical layer: hygienic design requirements covering surface finish, geometry, drainability, and cleanability.
Key 3A requirements:
- Surface finish: All product-contact surfaces must be finished to ≤ 32 micro-inch Ra (0.8 micrometer)
- No dead spaces: Seal geometry must prevent voids where bacteria can grow and CIP solutions cannot reach
- Drainability: Seals must allow complete drainage without product accumulation
- Material compatibility: Seals must withstand repeated CIP/SIP cycles without degradation
In January 2026, 3A SSI expanded its Replacement Parts and System Components Qualification Certificate program to grant the "RP Mark" to seals, gaskets, and O-rings. This applies particularly to rotary-shaft equipment, and the certification is mandatory for dairy processing and increasingly expected across food and beverage applications broadly.
USDA Compliance: Meat and Poultry Processing Requirements
Where 3A certification governs hygienic design, USDA compliance governs the meat and poultry sector specifically. For federally inspected facilities, USDA FSIS requires that compounds used on direct food-contact surfaces comply with the Federal Food, Drug, and Cosmetic Act (FFDCA) and applicable FDA regulations.
Documentation required: Suppliers must provide a Letter of Guaranty that includes:
- Statement of FFDCA compliance
- Specific product identification and lot traceability
- Intended use conditions (temperature, pressure, food types)
- Authorized signature from manufacturer
Holding multi-standard credentials (FDA + 3A + USDA) reduces procurement complexity and consolidates audit documentation. Artesian Systems stocks materials compliant with FDA 21 CFR 177.2600 and 177.1550, 3A-approved grades, and USDA standards, all managed under ISO9001 quality systems.
Types of PTFE Seals Used in Food Processing Equipment
Gaskets and O-Rings (Static Seals)
Static PTFE seals—flat gaskets, ring gaskets, and O-rings—are used in pipe flanges, sanitary tri-clamp fittings, vessel manways, and valve bodies. Virgin PTFE and expanded PTFE (ePTFE) gaskets are particularly suited to the low bolt-load, clamp-style connections common in sanitary piping systems.
Applications:
- Tri-clamp connections in dairy and beverage lines
- Flanged vessels and reactor manways
- Valve seats and pump body seals
- Heat exchanger plate gaskets
PTFE tri-clamp gaskets are standard in sanitary piping connections; they create leak-proof seals between stainless steel fittings without requiring high clamping forces.
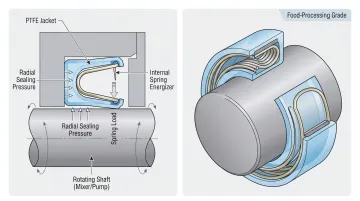
Lip Seals and Spring-Energized Seals (Dynamic Seals)
Dynamic PTFE seals serve rotating and reciprocating equipment—mixers, pumps, homogenizers, and filling heads. Spring-energized designs consist of a PTFE seal jacket with an internal spring energizer that applies continuous pressure to the seal lips.
Why springs are necessary: PTFE lacks elastic memory. Without an internal spring, a PTFE lip seal loses contact with a rotating shaft as the seal wears. Temperature-driven dimensional shifts compound this problem. The spring compensates for both variables, ensuring consistent sealing force throughout the seal's service life.
Key features:
- Temperature range: -268°C to 232°C (-450°F to 450°F)
- Self-lubricating (no external food-safe lubricant required)
- Zero water absorption for dimensional stability in steam environments
- Compliant with FDA 21 CFR Part 177, 3A Sanitary Standards 18-03 and 20-27, NSF/ANSI 2-2021
Dynamic PTFE seals are used extensively in cheese processing equipment—cookers, mixers, blenders, screw conveyors—where shaft speeds and aggressive washdown procedures would rapidly degrade standard elastomeric seals.
Encapsulated and Envelope Gaskets
Where dynamic seals handle movement, encapsulated gaskets solve a different problem: sealing irregular or low-load flanges that solid PTFE cannot reliably grip. Encapsulated (envelope) PTFE gaskets feature a PTFE jacket surrounding an elastomeric core—typically EPDM—delivering chemical inertness at the food-contact surface while the rubber core provides compressibility and elastic recovery.
Tuf-Flex unitized gaskets take this further by chemically bonding the PTFE jacket to the EPDM core—eliminating the separation failures common in standard envelope gaskets. The unitized construction delivers several advantages:
- Prevents creep failures inherent in solid PTFE gaskets during SIP cycles
- Maintains inner diameter without constant re-torquing
- Offers minimal thermal expansion compared to solid PTFE
Temperature rating: -29°C to 149°C (-20°F to 300°F). Compliant with FDA 21 CFR 177.2600 and 177.1550, USP Class VI, USDA, 3A Sanitary Standards, CGMP, and ASME-BPE.
Artesian Systems offers tri-clamp envelope gaskets (PTFE x EPDM) in sizes from 1.5" to 12", combining chemical resistance with enhanced sealing memory for applications where slight ferrule misalignment may occur.
Where FDA-Compliant PTFE Seals Are Used: Key Applications
Dairy, Beverage, and Liquid Food Processing
PTFE seals are the standard in milk pasteurizers, cheese processing equipment, beverage bottling lines, and juice processing systems. These applications expose seals to:
- Acids (citric, lactic, carbonic)
- Fats and dairy proteins
- Sugars and flavor compounds
- Frequent hot-water CIP cycles (65-80°C with 1-3% NaOH)
- Acid rinses (0.5-1.5% HNO₃ at 55-65°C)
PTFE rotary shaft seals in cheese cookers and mixers use FDA-approved PTFE rotor cups and stators, holding USDA certification for dairy, meat, and poultry along with 3A approval and EC Regulation 1935/2004 compliance. The seals maintain integrity through multiple daily CIP cycles that would cause elastomeric seals to swell, harden, or lose tensile strength.
Meat Processing, Dry Goods Filling, and High-Speed Portioning Equipment
In meat processing and high-speed filling operations, PTFE rotary shaft seals and lip seals must prevent lubricant migration into the food zone while withstanding high shaft speeds and aggressive washdown procedures. PTFE's self-lubricating properties (coefficient of friction 0.1) eliminate the need for external lubricants that could contaminate product.
That lubricant-free design also reduces a broader contamination risk: foreign material (including rubber gasket fragments) drove more recalls than E. coli in 2022, and PTFE's resistance to caustic washdown degradation directly limits that liability.

Pharmaceutical, Nutraceutical, and Biotech Processing
Sterile fluid transfer, reactor vessels, and biopharmaceutical blending equipment require seals that meet both FDA and USP-grade standards. PTFE's compliance with pharmaceutical regulations makes it well-suited for sterilization media including:
- Superheated steam (up to 150°C)
- Peracetic acid
- Hydrogen peroxide
Spring-energized PTFE seals maintain contact through thermal cycling between ambient production temperatures and steam sterilization at 121-134°C. Because PTFE absorbs essentially no water, seals don't swell or distort during repeated steam cycles — preserving a reliable, consistent seal across hundreds of sterilization runs.
CIP/SIP Compatibility: Surviving Aggressive Sanitation Cycles
CIP Parameters: Caustic, Acid, and Thermal Stress
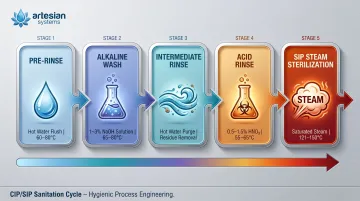
Clean-in-Place (CIP) cycles in dairy processing typically involve:
- Alkaline stage: 1-3% NaOH at 65-80°C to dissolve fat and protein residues
- Acid stage: 0.5-1.5% HNO₃ at 55-65°C to remove milk scale and calcium deposits
- Rinse cycles: Hot water at 60-80°C
- Frequency: Multiple times daily in dairy tanks and pasteurizers
SIP goes further — superheated steam at 121-134°C per pharmaceutical standards, with some systems reaching 150°C. Elastomeric seals routinely show hardness change, volume swell, and tensile strength loss under repeated exposure to both CIP and SIP conditions. PTFE's chemical inertness and thermal stability allow it to survive these cycles without progressive degradation.
Hygienic Design: Surface Finish and Dead Space Prevention
3A Sanitary Standards require all product-contact surfaces—including seal surfaces—to be finished to ≤ 32 micro-inch Ra (0.8 micrometer). A PTFE seal that traps food residue or cleaning solution in dead zones undermines the entire sanitation cycle, regardless of material compliance.
That surface finish requirement exists for a reason — dead spaces and voids behind seals are exactly where bacteria colonize and CIP solutions fail to penetrate. The January 2026 expansion of the 3A RP Mark to cover seal replacement parts directly addresses this risk. Proper sanitary seal design requires:
- Smooth surfaces without crevices
- Drainable geometry that prevents fluid accumulation
- Elimination of gaps between seal and mating surface
- Proper surface finish on both seal and equipment
Operational Benefits: Reduced Downtime and Audit Readiness
Meeting those design standards pays off on the production floor. PTFE seals that survive CIP/SIP cycles intact eliminate the unplanned stops that come with elastomeric seal failure or degradation. In practice, that means:
- Fewer unscheduled maintenance stops
- Reduced seal inventory and replacement labor
- Lower contamination risk from seal degradation or fragment shedding
- Simplified compliance documentation for regulatory audits
A seal that combines FDA material compliance, 3A hygienic design certification, and documented CIP/SIP test data covers multiple audit checkboxes with a single component — which matters when an FDA inspector or third-party auditor asks for documentation during a facility review.
How to Choose the Right FDA-Compliant PTFE Seal for Your Operation
Match Seal Type to Application Dynamics
Static connections (flanges, fittings, vessels, valve bodies) require gaskets or O-rings. Virgin PTFE and ePTFE gaskets excel in low bolt-load sanitary clamp connections.
Dynamic applications (rotating shafts, reciprocating pistons, pump impellers) require lip seals, spring-energized seals, or rotary shaft seals. Spring-energized designs are preferred because they compensate for PTFE's lack of elastic memory, maintaining seal contact despite wear, thermal cycling, and shaft misalignment.
Selecting the wrong type compromises both sealing performance and seal life—a static gasket design cannot maintain contact on a rotating shaft, and a dynamic lip seal may not distribute load properly in a bolted flange.
Determine Applicable Regulatory Standards
Food-contact surfaces in general food processing: FDA 21 CFR 177.1550 (virgin PTFE) or 177.2600 (compounded rubber/elastomeric articles).
Dairy operations: 3A sanitary standards typically required in addition to FDA compliance.
USDA-inspected meat and poultry facilities: FDA compliance documented via supplier Letter of Guaranty, plus adherence to FSIS guidelines.
Pharmaceutical and biopharmaceutical: FDA compliance plus USP Class VI certification for materials in sterile processing.
Confirm all applicable standards before specifying a seal. A seal that meets FDA material requirements but lacks 3A certification may fail a dairy audit, even though the material itself is compliant.
Virgin vs. Filled PTFE: Balancing Chemical Inertness and Mechanical Stability
| Property | Virgin PTFE | 25% Glass-Filled | 25% Carbon-Filled |
|---|---|---|---|
| Shore D Hardness | 50-65 | 60 | 62 |
| Tensile Strength | 3,000-5,000 psi | 2,100 psi | 1,900 psi |
| Creep Resistance | Low (cold flow) | Significantly improved | Significantly improved |
| FDA Compliance | Inherent (177.1550) | Requires filler verification | Requires filler verification |
| Chemical Resistance | Universal | Reduced (glass not suitable for strong alkalis/HF) | Good (carbon may affect purity) |

Virgin PTFE offers maximum chemical inertness and is the baseline FDA-compliant choice for food-contact surfaces. It is ideal for applications with low-to-moderate clamping loads and where re-torquing protocols can be implemented.
Filled PTFE grades improve creep resistance and dimensional stability under sustained load, making them suitable for high-pressure or high-temperature applications. However, any filled grade requires verification that the fillers themselves are FDA-compliant, and fillers may reduce chemical resistance to specific CIP agents (e.g., glass fillers are incompatible with hydrofluoric acid).
When cold-flow management is critical, EPDM-core encapsulated gaskets offer an alternative to solid virgin PTFE—combining PTFE's chemical inertness with a flexible core that maintains sealing load under compression.
Evaluate Operating Environment Parameters
Temperature range: Continuous exposure above 200°C or steam sterilization at 121-150°C requires high-temperature PTFE grades.
Chemical media: Identify both production media (food products) and sanitation agents (CIP chemicals). Verify PTFE compatibility with the most aggressive agent in the cycle.
Pressure: High-pressure applications (>100 psi) may require filled PTFE or envelope gaskets to prevent cold flow and leakage.
Shaft speed (dynamic seals): High-speed rotating equipment (mixers, homogenizers) requires spring-energized lip seals to maintain contact despite centrifugal forces.
CIP/SIP cycle frequency: Daily or multiple-daily cleaning cycles favor virgin PTFE or unitized envelope gaskets designed for repeated chemical exposure without degradation.
Prioritize Suppliers with Full Traceability and Engineering Support
A seal that is correctly specified on paper but manufactured without quality controls can still fail a compliance audit. Prioritize suppliers who provide:
- Full material traceability with lot numbers
- Documented FDA compliance certificates (specifying 177.1550 and/or 177.2600)
- 3A compliance documentation where applicable
- USDA Letter of Guaranty for meat and poultry applications
- ISO9001 or equivalent quality management certification
Artesian Systems supplies FDA 21 CFR 177.2600/1550 compliant, 3A-approved, and USDA-compliant sealing components under ISO9001 certification. With almost two decades of food and chemical processing experience, the company provides both off-the-shelf sanitary gaskets and custom-engineered seal solutions where standard sizes don't fit.
Frequently Asked Questions
What does FDA 21 CFR 177.1550 mean for PTFE seals?
21 CFR 177.1550 is the FDA regulation covering virgin (unfilled) PTFE as a material safe for repeated food contact. Seals carrying this compliance designation have been verified through extraction testing to ensure they do not leach harmful substances into food products, meeting extractive limits of ≤3.1 mg/sq dm total and ≤0.46 mg/sq dm fluoride.
Can PTFE seals withstand CIP and SIP cleaning procedures?
Yes. PTFE's chemical inertness and thermal stability (continuous service to 260°C) handle CIP caustic cleaners (1-3% NaOH), acid rinses (0.5-1.5% HNO₃), and SIP steam at 121-150°C without degradation. Seal geometry must also follow 3A hygienic design principles to prevent residue entrapment and ensure complete drainability.
What is the difference between virgin PTFE and filled PTFE in food processing?
Virgin PTFE is chemically pure and inherently FDA-compliant under 21 CFR 177.1550, but prone to cold flow under sustained load. Filled PTFE adds FDA-approved fillers (glass, carbon, bronze) for improved creep resistance—each filler must meet FDA standards independently and may reduce compatibility with some CIP chemicals.
Are PTFE seals compatible with 3A sanitary standards?
Yes, but compliance requires more than FDA-approved materials. 3A standards address hygienic design—surface finish (≤32 micro-inch Ra), drainability, crevice-free geometry, and cleanability. PTFE seals used in dairy and food processing should meet both FDA material requirements (177.1550/177.2600) and 3A design criteria. The January 2026 3A RP Mark for seal replacement parts specifically certifies this combined compliance.
How often do PTFE seals need to be replaced in food processing equipment?
Replacement frequency depends on application type, operating conditions (temperature, pressure, shaft speed), and CIP/SIP cycle intensity. Dynamic seals in high-speed or high-temperature service require regular inspection per the manufacturer's schedule. Static gaskets may need periodic re-torquing or replacement with envelope designs that resist cold flow.
What types of PTFE seals are most commonly used in dairy and beverage processing?
The three most common types are flat/ePTFE gaskets for tri-clamp and flange connections, spring-energized lip seals for pump shafts and mixer drives, and encapsulated (envelope) gaskets—a PTFE jacket over an EPDM core—for flanges with variable clamping loads or irregular surfaces.


